Starting with benzene and isopropyl chloride, show how you would prepare the following compound: N- -NH2 O,N-
Question:
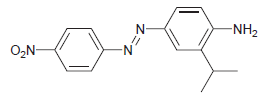
Transcribed Image Text:
N- -NH2 O,N-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
AICI 3 1 HNO3 HSO4 2 Fe HO ...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using toluene as your only source of carbon atoms, show how you would prepare the following compound.
-
Beginning with 3-methyl-1-butyne, show how you would prepare the following compounds: (a) (b) (c) Br CH3 CH2=C-CH-CH CH3 CH2Br--CBr2-CH-CH3 Br CH3 CH3-CH-CH CH
-
Starting with benzene and the appropriate acyl chloride or acid anhydride, outline a synthesis of each of the following: (a) Butylbenzene (b) (c) (d) Diphenylmethane 9,10-Dihydroanthracene
-
Assume that 3-month Treasury bills totaling $23 billion were sold in $10,000 denominations at a discount rate of 5.200%. In addition, the Treasury Department sold 6-month bills totaling $21 billion...
-
If XYZ Corp. can undertake the following projects: Project 1: Required investment: $10 million Expected rate of return: 12% Project 2: Required investment: $2 million Expected rate of return: 15%...
-
1. A & F defenders have argued that the clothier has the right to promote an image of its choice as a means of making a profit. a. The law aside, is Abercrombie simply wrong to project an image that,...
-
A consumer advocacy agency, Equitable Ernest, is interested in providing a service that allows an individual to estimate their own credit score (a continuous measure used by banks, insurance...
-
Following is Hartzbergs latest income statement. The company produced and sold 100,000 units during the year. Required Restate the income statement in contribution margin format, as if Hartzburg had...
-
How does the application of advanced statistical mechanics and quantum thermodynamics contribute to a deeper understanding of phase transitions and critical phenomena in complex systems, particularly...
-
A 400 N sphere is resting in a trough as shown in fig. 1. Determine the reactions developed at contact surface. 60 450 Fig.1
-
Identify the reactants you would use to prepare each of the following azo dyes via an azo coupling reaction: (a) (b) (c) N- !i -NH2 S SO,H -NO2
-
Draw the product obtained when the diazonium salt formed from aniline is treated with each of the following compounds: (a) Aniline (b) Phenol (c) Anisole (methoxybenzene)
-
Describe how the American Association of Law Librarians recommends that a person cite information obtained from the Internet.
-
The Foodpanda group is a worldwide mobile food delivery marketplace headquartered in Berlin, Germany. It was launched in March 2012. It allows you to select from local restaurants and place orders...
-
What is the formula for the rotational inertia of a thin spherical shell of inertia \(m\) and radius \(R\) about an axis tangent to any point on its surface?
-
For a given level of technology, there is a maximum density of "bits" of information that can be stored on a computer's hard-drive disk. The data are stored in a tight spiral, so that "tracks" are...
-
A gymnast's backflip is considered more difficult to do in the layout (straight body) position than in the tucked position. Why?
-
You have a pail of water with a rope tied to the handle. If you whirl the pail in a vertical circle fast enough, none of the water spills out, even when the pail is upside down. Explain why.
-
Graph each function in the viewing window specified. Compare the graph to the one shown in the answer section of this text. Then use the graph to find (1.25). (x) = (3x - 1)(x + 2) 2 ; window: [-4,...
-
Air pollution generated by a steel mill is an example of a) a positive production externality. b) a negative production externality. c) a public good. d) the free-rider problem. State and local taxes...
-
What do the pericyclic selection rules have to say about the position of equilibrium in each of the reactions given in Fig. P27.30? Which side of each equilibrium is favored and why? Fig. P27.30 (a)...
-
What stereoisomer of A also gives compound C on heating?
-
Suppose the migrating methyl group in part (a) were labeled with the hydrogen isotopes deuterium (D) and tritium (T) so that it is a -CHDT group with the S configuration. What would be the...
-
A bank can either invest money for three months at 4.00% or for nine months at 4.50%. Ignoring actual/360 day count adjustments for the purpose of this question, the three against nine FRA quote the...
-
Shuggy Otis, an executive at Slapfish Corp. (SC) intends to retire in 11 years. SC just announced that it will start depositing $500.00 at the end of each quarter into each of its workers' retirement...
-
On January 1, the Hanover Beverage Company replaced the palletizing machine on one of its juice lines. The cost of the machine was $195,000. The machine's expected life is five years or 480,000...

Study smarter with the SolutionInn App


