Starting with benzene and using any other necessary reagents of your choice, design a synthesis for each
Question:
Starting with benzene and using any other necessary reagents of your choice, design a synthesis for each of the following compounds:
(a)
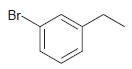
(b)
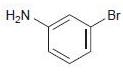
(c)
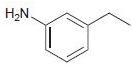
Transcribed Image Text:
Br-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a b c 1 CH...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Starting with benzene and using any other reagents of your choice, show how you would prepare each of the following compounds: a. b. c. d.
-
Outline a synthesis for each of the following compounds from the indicated starting material and any other reagents. (a) l-chloro-3, 5-dinitrobenzene from benzene (b) 2-chloro-4,6-dinitrophenol from...
-
Starting with cyclopentanone and using any other reagents of your choosing, identify how you would prepare each of the following compounds: (a) (b) (c) (d)
-
Use the Chain Rule to calculate d/dt (r(t)) at the value of t given. f(x, y) = x = xy, r(t) = (R R - 41), t = 4
-
What can business owners do to maintain high ethical standards in their companies?
-
On June 1, 2014, Kappa Company acquired all of the net assets of Delta Company for $ 120,000 cash. The statements of financial position for both companies prior to the acquisition follow: Required:...
-
The altitudes (in kilometers) of atmosphere at which helium is found in majority in 10 different cities are listed. 938.5 927.0 929.5 930.3 934.3 936.0 926.2 930.5 924.8 870.7 (a) Find the range of...
-
Selected pension information extracted from the retirement benefits note that appeared in Greens 2014 annual report follows. (These numbers have been modified but are based on the activities of a...
-
Use the excel in the link to create a detailed scope of work of a three-storey residential building. Note: No need to fill the Quantity & Unit Cost. Please see attached link:...
-
Figure a, shows a uniform beam subject to a linearly increasing distributed load. The equation for the resulting elastic curve is (see Figure b) Use bisection to determine the point of maximum...
-
How do project layouts differ from product- and process-focused layouts?
-
What is visual merchandising and micro-merchandising?
-
Brin Company issues bonds with a par value of $800,000. The bonds mature in 10 years and pay 6% annual interest in semiannual payments. The annual market rate for the bonds is 8%. 1. Compute the...
-
How does a crime compare to a civil wrong?
-
What is lenity? How does it place limitations on the criminal law?
-
Explain the principle of legality.
-
How do the separation of powers and federalism place limitations on governments law-making authority? Be specific.
-
Explain the relationship (real and ideal) between higher and lower courts.
-
To perform the division using synthetic division, we begin by writing the following. 3 x + 2)x + 4x + 2
-
What is an insurable interest? Why is it important?
-
In its latest catalog, Blarneystyne, Inc., a chemical company of dubious reputation specializing in alkynes, has offered some compounds for sale under the following names. Although each name...
-
Build a model of cyclodecyne. Compare its stability qualitatively to that of cyclohexyne; explain your answer.
-
On the basis of the hybrid orbitals involved in the bonds, arrange the bonds in each of the following sets in order of increasing length. C--C single bond of propan; C--C single bond of propyne; C--C...
-
In 2019, Rylan Enterprises' net income increased by $2.5 million while its depreciation expense decreased by $500,000, accounts receivable increased by $2,000,000 and accounts payable increased by...
-
Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company has two manufacturing departments-Molding and Fabrication. It started, completed, and sold...
-
The Sky Blue Corporation has the following adjusted trial balance at December 31. Cash Accounts Receivable Prepaid Insurance Notes Receivable (long-term) Equipment Accumulated Depreciation Accounts...

Study smarter with the SolutionInn App


