The following mass spectrum is for octane. a) Which peak represents the molecular ion? b) Which peak
Question:
a) Which peak represents the molecular ion?
b) Which peak is the base peak?
c) Draw the structure of the fragment that produces the base peak.
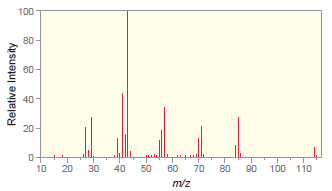
Transcribed Image Text:
100 80 60 40 20 110 10 20 30 40 50 60 70 80 90 100 m/z Relative Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
a The molecular io...View the full answer

Answered By

Nandana Wijayarathna
I am a highly experienced writer in several areas,
Business management
Information technology
Business administration
Literature
Biology
Environmental science
History
4.50+
161+ Reviews
399+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
2-Methylpentanc (C 6 H 14 ) has the mass spectrum shown. Which peak represents M + ? Which is the base peak? Propose structures for fragment ions of m/z = 71, 57, 43, and 29. Why does the base peak...
-
Calculate the mass of octane, C8H18(l), that must be burned in air to evolve the same quantity of energy as produced by the fusion of 1.0 g of hydrogen in the following fusion reaction: Assume that...
-
Use Figure 24-15 to suggest which type of liquid chromatography you could use to separate compounds in each of the following categories: (a) Molecular mass < 2 000, soluble in octane (b) Molecular...
-
In Exercises 7980, find the value of y if the line through the two given points is to have the indicated slope. (3, y) and (1, 4), m = -3
-
Write an essay about a "Magazine" focusing on the demographics, describing the niches the magazine is trying to reach. For this the chosen magazine is: Elle magazine. Some demographics that should be...
-
The file Property Taxes contains the property taxes per capita for the 50 states and the District of Columbia. Decide whether the data appear to be approximately normally distributed by a. Comparing...
-
The composite pressure vessel in Problem 8.6 is subjected to an internal pressure that varies sinusoidally with time according to the relationship \(p(t)=P_{0}\) \(\sin t\), and the principal complex...
-
Using the financial information presented in Exhibit 13.1, calculate the following ratios for The Home Depot: Return on equity Return on assets Total asset turnover Inventory turnover Current...
-
What is relationship between the various sides of the Iron Triangle? Do you believe that it is possible to have a balance of the triangle wherein the vast majority of users are satisfied? Discuss why...
-
Verify that (a) (2 - i) - i(1 - 2i) = -2i; (b) (2, - 3)(-2, 1) = (-1, 8) (c) (3, 1)(3-1)( TO 5 10 .
-
Propose a molecular formula that fits the following data. a) A hydrocarbon (C x H y ) with a molecular ion peak at m/z = 66 b) A compound that absorbs IR radiation at 1720 cm -1 and exhibits a...
-
Calculate the HDI for each molecular formula. a) C 4 H 6 b) C 5 H 8 c) C 40 H 78 d) C 72 H 74 e) C 6 H 6 O 2 f) C 7 H 9 NO 2 g) C 8 H 10 N 2 O h) C 5 H 7 Cl 3 i) C 6 H 5 Br j) C 6 H 12 O 6
-
Refer to Campbell Soup Companys statement of cash flows in Appendix A. Required: Convert Campbells statement of cash flows for Year 11 to show cash flows from operations (CFO) using the direct...
-
What are some motives for divestitures?
-
What are the three types of risk to which projects are exposed? Which type of risk is theoretically the most relevant? Why?
-
Differentiate between spot and forward exchange rates.
-
Describe the pure play and the accounting beta methods for estimating divisional betas.
-
Explain the reasoning behind the bond-yield-plus-judgmental-risk premium approach.
-
Graph each polynomial function. Factor first if the polynomial is not in factored form. (x) = x 2 (x - 2)(x + 3) 2
-
The outer loop controls the number of students. Note that the inner loop of this program is always executed exactly three times, once for each day of the long weekend. Modify the code so that the...
-
Is there an unbranched alkane containing 23 hydrogen atoms? If so, give its structural formula; if not, explain why not.
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
1. Define latent heat and how it is different than specific heat capacity. 2. Describe how a phase diagram changes when changing from a solid to a liquid. 3. Describe how work done is related to a...
-
In a large vaccination clinic, patients arrive at the rate of 50 per hour. The clinic is staffed with five nurses and it takes on average 6 minutes for a nurse to vaccinate a patient. Both patient...
-
Calculate the missing value. Beginning cash balance add : cash receipts Collection of notes receivable Proceeds from sale of securities collection from credit sales Total receipts Total available...

Study smarter with the SolutionInn App


