Using acetophenone as your only source of carbon atoms, propose a synthesis for the following compound. Acetophenone
Question:
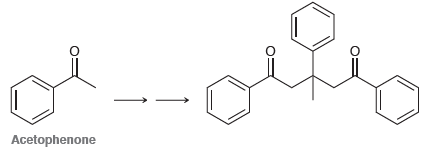
Transcribed Image Text:
Acetophenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
RNH H HO ...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using benzene as your only source of carbon atoms and ammonia as your only source of nitrogen atoms, propose a synthesis for the following compound: HN NH
-
Using acetylene as your only source of carbon atoms, outline a synthesis for 3-hexyne.
-
Using acetylene as your only source of carbon atoms, design a synthesis of cis-3- decene:
-
During the year ended 30 June 20X5, Gem Stone Ltd acquired two areas of interest Site A and Site B. The exploration and evaluation activities at Site A amounted to $64454401. The costs relate to the...
-
An adjustable rate mortgage with a teaser start rate of 1.5%, an index of six month LIBOR, a margin of 2.0%, and periodic and lifetime caps of 2/6 with semi-annual adjustments, would have a maximum...
-
For each of the basic blocks of Q5-4 , determine the order of execution of operations that gives the smallest number of required registers. Next, state the number of registers required in each case....
-
Describe the role of the patient, physician, nurse, and hospital in obtaining informed consent.
-
I recently sold my used car. If no new production occurred for this transaction, how could it have created value?
-
Your company is excited with the information discussed regarding the cloud technologies that are available to Get-Sole Shoes. Now the management team wants to examine what its cloud infrastructure...
-
Draw two sets of axes, one for Nation 1 and the other for Nation 2, measuring labor along the horizontal axis and capital along the vertical axis. (a) Show by straight lines through the origin that...
-
Assign groups of students to go to an airport (or other location where they will find lots of foot traffic) to people watch. Have them classify as many people as possible according to social class...
-
Draw a complete mechanism for the following transformation. NaOH, heat
-
For (x) = 3x and g(x) = [1/4] x , find each of the following. Round answers to the nearest thousandth as needed. (-1.68)
-
A businessperson you know has just received the financial statements of a company in which that person owns shares. Answer the following questions asked by the person. Try to answer without jargon,...
-
What is the role of information in a capital market?
-
North Shore Manufacturing Ltd had this balance sheet: During July 2016, North Shore Manufacturing experienced the following transactions: a. An amount of $8000 of accounts payable was paid, b. A...
-
On 1 January 2016, Y Ltd acquired 100 000 shares (30 percent of the voting interest) in P Ltd for $900 000 cash. On 30 June 2016, P Ltd announced its earnings per share for the first six months of...
-
The balance sheet and income statement of Reconstruction Limited are reproduced below. 1. How much cash was paid for insurance during the year? 2. How much cash was paid for electricity during the...
-
Show that 9300 cal of heat is needed to change 15 g of 20C water to 100C steam.
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
Give the principal organic product(s) expected when propionyl chloride reacts with each of the following reagents. (a) H2O (b) Ethanethiol, pyridine, 0 C (c) (CH3)3COH, pyridine (d) (CH3)2CuLi, -78...
-
Give the structure of a compound that satisfies each of the following criteria. (a) A compound C3H7ON that liberates ammonia on treatment with hot aqueous KOH (b) A compound that gives equal amounts...
-
When (R)-(-)-mandelic acid (-hydroxy--phenylacetic acid) is treated with CH3OH and H2SO4, and the resulting compound is treated with excess LiAlH4 in ether, then H2O, a levorotatory product is...
-
A 1 kg metal block is heated to 200 C and then dropped into a thermally isolated container with 4 kg of water and 100 gr of ice both at a temperature of 0 C. If the specific heat of water is cw=4186...
-
Why are both ER Diagrams and Relational Modeling needed when a database system is developed?
-
Which data model provides a user with the best "program-data independence"? A. Hierarchial Model B. Network Model C. Relational Model D. Entity Relationship Model Why?

Study smarter with the SolutionInn App


