Using benzene as your only source of carbon atoms and ammonia as your only source of nitrogen
Question:
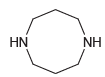
Transcribed Image Text:
HN NH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Na NH3 CH3OH ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a plausible synthesis for 1,4-dioxane using acetylene as your only source of carbon atoms. 1,4-Dioxane
-
Starting with 1,3-butadiene as your only source of carbon atoms, and using any other reagents of your choice, design a synthesis of the following compound:
-
Using ethanol as your only source of carbon atoms, design a synthesis for the following compound:
-
On 28 April 2020, Mr Guna, CEO of Econ Engineering Malaysia, proposed to complete an abandoned boiler project that no one had dared to revive. He knew that the project was 60% complete before it was...
-
1. An ________ is any happening for which the result is uncertain, and the possible results are called ________. 2. The set of all possible outcomes of an experiment is the ________ ________. 3. The...
-
Is higher Medicaid cost-sharing a good idea? What are the strongest arguments you can make for and against higher cost sharing? Should Medicaid beneficiaries have the same cost sharing...
-
In Section 2.3, 20 patients for Example 2.5 were randomly selected from a population of 25,000 . a. Explain why the probability of choosing a random sample of 20 from 25,000 is...
-
As she headed toward her boss's office, Emily Hamilton, chief operating officer for the Aerocomp Corporation-a computer services firm that specialized in airborne support-wished she could remember...
-
Use the information provided in the table to answer the following questions. Assume that there were no changes in any other asset or liability accounts. December 2022 December 2023 Net Income...
-
Pick three different industries and environments. What different strategy development styles (classical, adaptive, shaping or visionary) would be appropriate in them?
-
Propose a synthesis for the following transformation: 'N'
-
Draw the structure of the compound with molecular formula C8H11N that exhibits the following 1 H NMR and 13 C NMR spectra: Proton NMR 2 22 Chemical Shift (ppm) Carbon NMR 128.8 128.4 40.0 -126.1...
-
Jackson Company recently calculated its break-even sales revenue to be $24,000. For each dollar of sales revenue, $0.60 goes to cover variable costs. Compute the following: a. The contribution margin...
-
Assume that, in 2016, ABB purchased a new automation technology for SFr500 million. They paid this on credit and wont be due to actually pay for the automation technology until 2018. The managers of...
-
Suppose you own shares in a company. The current share price is 2.50. Another company has just announced that it wants to buy your company and will pay 3.50 per share to acquire all the outstanding...
-
In recent years, governments have taken control of banks through buying their shares. What impact does this have on the lending culture of these banks? Is this consistent with shareholder...
-
Firm A and Firm B have debttotal asset ratios of 70 per cent and 30 per cent, and returns on total assets of 20 per cent and 30 per cent, respectively. Which firm has a greater return on equity?
-
Mill Co., a calendar-year manufacturer, which uses the FIFO inventory method, previously allocated production costs to inventory by use of a burden rate based on the ratios oft otal indirect...
-
A proton with momentum p 1 = 3 GeV elastically diffuses on a proton at rest. The diffusion angle of one of the protons in the CM is * ac = 10. Find, (a) The kinematic quantities in the L frame; (b)...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
All of the following names are incorrect or incomplete. In each case, draw the structure (or a possible structure) and name it correctly. (a) 2-methylethylpentane (b) 2-ethyl-3-methylpentane (c)...
-
Give structures and names for: (a) The five isomers of C6H14 (b) The nine isomers of C7H16
-
Draw the structures of the following groups, and give their more common names. (a) The (1-methylethyl) group (b) The (2-methylpropyl) group (c) The (1-methylpropyl) group (d) The (1,1-dimethylethyl)...
-
1) Based on the stock chart for Michaels Companies Inc, what do you think the short and long-term growth potentials are for this company? (discuss the advantages/disadvantages) Link to the stock...
-
After being drafted in the first round of the NFL draft, a star defensive end invests his signing bonus of $9,827,000.00 in a mutual fund. The fund pays on average 7.00% APR. The player will not...
-
XYZ Co has plans to issue 7,000, Eleven percent Debentures of Rs.100 each at a discount of 5%. The debentures are redeemable after 4 years and the commission payable to brokers & underwriters is Rs....

Study smarter with the SolutionInn App


