A simulated infrared absorption spectrum of a gas phase organic compound is shown in the following figure.
Question:
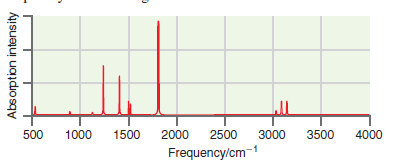
Transcribed Image Text:
1000 2000 500 1500 2500 3000 3500 4000 Frequency/cm-1 Absorption intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The peaks near 3100 cm 1 are indicative of CH stretching modes the peak ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
According to the Statute of Frauds, in order to be legally enforceable, a contract must be in writing, name the contracting parties, identify the subject matter of the contract, and Be for a legal...
-
In Exercises 1-4, use the half-angle formulas to determine the exact values of the sine, cosine, and tangent of the angle. 1. 75 2. 165 3. 112 30o 4. 67 330o
-
A football is thrown on a long pass. Compared to the balls initial vertical velocity, the vertical component of its velocity at the highest point is (a) greater, (b) less, (c) the same.
-
Prove directly that, for each \(t \geq 0\), \[\mathbb{E}\left(e^{-z T(t)} ight)=\int_{0}^{\infty} e^{-z s} f_{T(t)}(s) d s=e^{-t z^{1 / 2}}\] where \((T(t), t \geq 0)\) is the Lvy subordinator.
-
From the information in BE5-3, prepare the journal entries to record these transactions on Rodriguez Companys books under a perpetual inventory system.
-
Public Company acquired the net assets of Sun Company by paying cash $420,000. At that time, the book value of the net assets of Sun Company was $300,000 and the fair value its net assets was...
-
Design and implement a better Web page for translating English text into Pirate talk. The JavaScript code for performing the translation is provided below. The PHRASES array contains English...
-
Purification of water for drinking using UV light is a viable way to provide potable water in many areas of the world. Experimentally, the decrease in UV light of wavelength 250 nm follows the...
-
The molecules 16 O 12 C 32 S and 16 O 12 C 34 S have values for h/8Ï 2 I of 6081.490 Ã 10 6 s 1 and 5932.816 Ã 10 6 s 1 , respectively. Calculate the CO and CS bond distances. The...
-
Companies that only import and/or export to operate globally. Identify each practice as more likely to be used by large or small global companies: A. Large MNCs B. Small international companies
-
How does the dynamic capabilities framework improve upon the resource-based view of strategy?
-
What are the advantages and disadvantages of following a multidomestic strategy?
-
First-mover advantages are more important than a companys capabilities. Discuss.
-
How does strategic group analysis enable a company to understand its competitors?
-
What is a combination lease?
-
The price p, in dollars, of a certain product and the quantity x sold obey the demand equation Suppose that the cost C, in dollars, of producing x units is Assuming that all items produced are sold,...
-
Tarick Toys Company manufactures video game consoles and accounts for product costs using process costing. The following information is available regarding its June inventories. The following...
-
Draw an MO energy diagram and determine the bond order for O 2 2 -. Is it diamagnetic or paramagnetic?
-
A polar molecule will exhibit dipoledipole forces. Which molecules will show dipoledipole forces? a) CHF 3 b) OCS c) H 2 S
-
Calculate the energy in kJ necessary to vaporize 100 g of water at its boiling point.
-
1. Develop a definition for the Triple C model of project management. 2. List some of the factors that can impede the flow of information for project planning purposes. How can these factors be...
-
The Meat Mart has $900,000 in net income. The firm has 200,000 shares of stock outstanding. The market price per share is $76. What is the PE (price to earnings) ratio?
-
Cost of Goods Manufactured for a Manufacturing Company The following information is available for Fuller Manufacturing Company for the month ending January 31: Cost of direct materials used in...

Study smarter with the SolutionInn App


