An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic
Question:
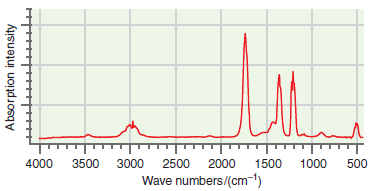
Transcribed Image Text:
T. 4000 3500 3000 2500 2000 1500 1000 500 Wave numbers/(cm-1) Absorption intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The major peak near 1700 cm 1 is the CO stretch and the peak nea...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The market for oil-based paint is shown in the following table: Suppose the production of the paint creates a negative externality of $10 for each unit of paint, which is the cost of repairing the...
-
Use the following information to decide whether this equipment lease qualifies as an operating, sales-type, or direct financing lease to a lessor. (a) There is no transfer of ownership at the end of...
-
The part shown in the following figure is a carbon-steel segment (partial) gear. The smaller hole at the bottom is for clamping the part onto a round shaft, using a screw and a nut. Suggest a...
-
An important U.S. government organization charged with setting human resource management guidelines is O the EEOC (Equal Employment Opportunity Commission). the OSHA (Occupational Safety and Health...
-
In Exercises 1-2, use the given conditions to find the exact values of sin 2u, cos 2u, and tan 2u using the double-angle formulas. 1. sin u = 3 / 5, 3 / 2 < u < 2 2. cos u = 4 / 5, / 2 < u <
-
Which of the following scenarios illustrates the concept of voluntary jurisdiction? (More than one answer might be literally true. Choose the best one.) a. An out-of-state defendant who signs a...
-
Show that the stochastic process \[e^{\int_{0}^{t} c(s) d B_{s}-\frac{1}{2} \int_{0}^{t} c^{2}(s) d s}\] is a martingale for any deterministic function \(c(t)\). Does the result change if \(c(t,...
-
The standard operating capacity of Tecate Manufacturing Co. is 1,000 units. A detailed study of the manufacturing data relating to the standard production cost of one product revealed the following:...
-
what is the amount of the difference between the variable costing and absorption costing net operating income (losses) If the sales volumes in the east and west regions have been reversed what would...
-
The Bombay Beverage Company has heavily advertised its social responsibility to the environment and community. In its advertisements, the company touts how its recycling efforts have reduced its...
-
The rotational constant for 14 N 2 determined from microwave spectroscopy is 1.99824 cm 1 . Calculate the bond length in 14 N 2 to the maximum number of significant figures consistent with this...
-
Calculate the zero point energies for 1 H 19 F and 2 D 19 F. Compare the difference in the zero point energies to k B T at 298 K.
-
Draw Bode diagram for unity feedback system \[ \mathrm{G}(s)=\frac{\mathrm{K}}{s(s+2)(s+10)} \] Read as best you can, the value of \(\omega_{p c}\). For what value of \(\mathrm{K}\) will \(\omega_{p...
-
Identify the capabilities that reside inside the Tata Group. To what extent do these capabilities provide Tata with a competitive advantage?
-
What are the three types of project risk?
-
How is each type of project risk measured?
-
Joint ventures are not an effective way to share capabilities between companies. Discuss.
-
In 2005 Nike returned to reporting on its social and environmental practices after a couple of years of silence due to legal concerns. The sports and clothing company is very important to countries...
-
(a) The CPI was 163.0 for 1998 and 215.3 for 2008. Assuming that annual inflation remained constant for this time period, determine the average annual inflation rate. (b) Using the inflation rate...
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
Consider the reaction: 2 COF 2 (g) CO 2 (g) + CF 4 (g) Kc = 2.00 In an equilibrium mixture, the concentration of COF 2 is 0.35 M and the concentration of CO 2 is 0.144 M. What is the equilibrium...
-
Consider the reaction: N 2 O 4 (g) 2 NO 2 (g) Kc = 0.36 A reaction mixture initially contains [N 2 O 4 ] = 0.100 M. Find the equilibrium concentrations of N 2 O 4 and NO 2 .
-
Consider the reaction: 2 H 2 S (g) 2 H 2 (g) + S 2 (g) Kc = 1.67 10 -7 A reaction mixture initially contains [H 2 S] = 0.010 M. Find the equilibrium concentrations of H 2 and S 2 .
-
The controller for Tulsa Medical Supply Company has established the following activity cost pools and cost drivers. Machine setups Budgeted Overhead Cost Cost Driver Number of setups Weight of raw...
-
In 2023, Miranda records net earnings from self-employment of $168,500. She has no other income. Determine the amount of Miranda's self-employment tax and her AGI income tax deduction. In your...
-
Describe how empowerment, work groups, and multifunctional teams would or would not affect the five types of problems.

Study smarter with the SolutionInn App


