Consider the following mechanism, which results in the formation of product P: If only the species A
Question:
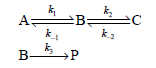
If only the species A is present at t = 0, what is the expression for the concentration of P as a function of time? You can apply the preequilibrium approximation in deriving your answer.
Transcribed Image Text:
k, ec B: A: k1 ka →P B-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Using the preequilibrium approximation we can express B and C ...View the full answer

Answered By

Omar ELmoursi
I'm Omar, I have Bachelor degree in Business and Finance, My unique approach is to help students with questions and assignments, I can teach Business, Math, Accounting, Managerial Accounting, Economy, Human resources management, organizational behavior, project management, I have experience dealing with different types of students and teach them how to deal with different types of exercises.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following mechanism for renaturation of a double helix from its strands A and B: Derive the rate equation for the formation of the double helix and express the rate constant of the...
-
Consider the following mechanism for the thermal decomposition ofR2: Where R2, PA' PB are stable hydrocarbons and Rand R' are radicals. Find the dependence of the rate of decomposition of R, on the...
-
Consider the following mechanism for a reaction in aqueous solution and indicate the species acting as a catalyst: Explain why you believe this species is a catalyst. What is the overall reaction?...
-
Question #1 The following table indicates the net cash flows of a capital asset: Year Net Cash Flow 0 $-8,400 1 $5,900 2 $6,200 Do not enter dollar signs or commas in the input boxes. Use the...
-
Determine whether the statement is true or false. Justify your answer. 1. If f is an even function, then f1 exists. 2. If the inverse function of f exists and the graph of f has a y-intercept, then...
-
(L03) In Year 1, Noah sells a collector automobile he purchased several years ago with a basis of \(\( \$ \) 20,000\) to a buyer for \(\( \$ \) 100,000\). As part of the purchase price, the buyer...
-
Consider the regression model (8.8) described in Example 8.3 Graph the response function for this model and indicate the role the model parameters play in determining the shape of this function....
-
SWAT Team Surplus began July with 66 tents that cost $23 each. During the month, SWAT Team Surplus made the following purchases at cost: Jul 4 105 tents @ $25 = $2,625 19 157 tents @ $27 = 4,239 25...
-
Sit Down Publishers publishes The Accountant magazine and sells one - year ddsubscriptions to its customers. Customers are required to pay in full for the one - year subscription at the beginning of...
-
Estimate the maximum diameter to which a thin-walled spherical shell can be expanded.If the diameter of the shell prior to expansion is 180mm and material have an allowable strain of 35 percentage A...
-
Consider the gas-phase isomerization of cyclopropane. Are the following data of the observed rate constant as a function of pressure consistent with the Lindemann mechanism? CH, CH,CH=CH2 CH,-CH2 k...
-
Using the preequilibrium approximation, derive the predicted rate law expression for the following mechanism: A+B- P
-
Suppose that there are n people in society, arranged (without loss of generality) in increasing order of income earned. Let x = (x 1 , . . . , x n) and y = (y 1 , . . . , y n ) be two income...
-
A bus is bought for 60,000. It will be used for four years, and then sold back to the supplier for 15,000. Show the depreciation calculations for each year using (a) the reducing balance method at...
-
After brainstorming, two enterprising college students have come up with what they think is the perfect personal protection app. One of these entrepreneurs, an engineering major, has designed a cell...
-
Why do most bond trades occur in the over-the-counter (OTC) market?
-
Do bond ratings adjust immediately to changes in credit quality? Explain.
-
Why are bond ratings important to firms and investors?
-
Firm HR is about to implement an aggressive long-term strategy consisting of three phases. It is crucial to the success of the strategy that the IRS accepts Firm HRs interpretation of the tax...
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
a. Calculate S if 1.00 mol of liquid water is heated from 0.00C to 10.0C under constant pressure if C P,m = 75.3 J K -1 mol -1 . b. The melting point of water at the pressure of interest is 0.00C and...
-
Show what reagents you would use to prepare each of the following ethers via an alkoxymercuration-demercuration. a. b. c. d. OEt
-
A 2.25 mole sample of carbon dioxide, for which C P,m = 37.1 J K -1 mol -1 at 298 K, is expanded reversibly and adiabatically from a volume of 4.50 L and a temperature of 298 K to a final volume of...
-
How does implementing Total Quality Management (TQM) contribute to enhancing both product and service quality within an organization?
-
What is the current gdp of India in 2023? difference between 2021-2023. please share all details in index form
-
Below you are provided with details of two companies and their budgets. Prepare the budgets and complete the following tasks: A. From the following information relating to Marmax Consulting Services,...

Study smarter with the SolutionInn App


