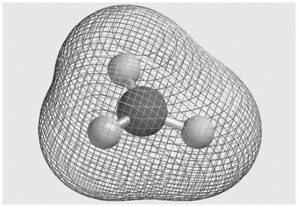
Consider the molecular electrostatic potential map for the BH 3 molecule shown here. Is the hydrogen atom
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
It is an electron acceptor becaus...View the full answer

Answered By

Omar ELmoursi
I'm Omar, I have Bachelor degree in Business and Finance, My unique approach is to help students with questions and assignments, I can teach Business, Math, Accounting, Managerial Accounting, Economy, Human resources management, organizational behavior, project management, I have experience dealing with different types of students and teach them how to deal with different types of exercises.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the molecular electrostatic potential map for the BeH 2 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the NH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the LiH molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
There are several types of interviews. List and define the methods of interviewing. List and discuss the different types of employment tests. List and discuss the uses of the performance appraisal -...
-
In Exercises 1-4, use an inverse trigonometric function to write as a function of x. 1. 2. 3. 4. 4 . 4
-
What is trade discount and what is meant by the successive way of calculating the multiple discount? Give a numerical example to illustrate your answer.
-
Explain the difference between strong and weak methods in Artificial Intelligence. Explain how this dichotomy differs from the difference between strong and weak AI.
-
The Runabout Boat Company is considering the purchase of several units of an expensive new machine for its production facilities around the country. Before committing to this purchase, Runabout wants...
-
Do you think McDonald's entry strategy was appropriate for the Indian market? Explain there strategy according to Indian market.
-
Raptor Fuels produces three grades of gasoline Regular, Premium, and Super. All of these are produced by blending two types of crude oil Crude A and Crude B. The two types of crude contain specific...
-
Distinguish between the following concepts used to describe chemical bond formation: basis set, minimal basis set, atomic orbital, molecular orbital, and molecular wave function.
-
Using Figures 23.9 and 23.10, explain why Î Ï 2 g < 0 and ÎÏ 2 u > 0 outside of the bonding region of H + 2 . Figure 23.9 Figure 23.10 Bonding - Antibonding . , Bonding...
-
Refer to the information for Irons Delivery Inc. above. Irons uses the straight-line method of depreciation. Irons Delivery Inc. purchased a new delivery truck for $45,000 on January 1, 2011. The...
-
5. Grace chooses a camel to ride on a carousel. The camel is located 9m from the centre of the carousel. If the carousel turns through an angle of determine the length of the are travelled by the...
-
How do ecological interactions, such as predation, competition, and mutualism, influence population dynamics and community structure in terrestrial and aquatic ecosystems ?
-
What are the molecular mechanisms underlying the process of cellular differentiation in multicellular organisms ?
-
If u(x) = x + 3 and v(x) = x - 9. For which value(s) of x is y = u(x) undefined?
-
How do cells communicate through gap junctions and adherens junctions? What roles do cadherins and integrins play in maintaining tissue structure and cell adhesion ?
-
How many terms must be added in an arithmetic sequence whose first term is 11 and whose common difference is 3 to obtain a sum of 1092?
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
Which of the following molecules may show infrared absorption spectra: (a) H 2 , (b) HCl, (c) CO 2 , (d) H 2 O?
-
The first five vibrational energy levels of HCl are at 1481.86, 4367.50, 7149.04, 9826.48, and 12 399.8 cm 1 . Calculate the dissociation energy of the molecule in reciprocal centimetres and...
-
For 16 O 2 , G values for the transitions v = 1 0, 2 0, and 3 0 are, respectively, 1556.22, 3088.28, and 4596.21 cm 1 . Calculate and x e . Assume ye to be zero.
-
Martin Company applies manufacturing overhead based on direct labor hours. Information concerning manufacturing overhead and labor for the year is as follows: Actual manufacturing overhead Estimated...
-
$ 719,000 280,000 439,000 150,400 Joyner Company's income statement for Year 2 follows: Sales Cost of goods sold Gross margin Selling and administrative expenses Net operating income Nonoperating...
-
FIFO and LIFO costs under perpetual inventory system The following units of an item were available for sale during the year: Beginning inventory 22 units at $44 Sale 18 units at $62 First purchase 18...

Study smarter with the SolutionInn App


