Cyclopropane isomerizes into propene when heated to 500C in the gas phase. The extent of conversion for
Question:
Cyclopropane isomerizes into propene when heated to 500°C in the gas phase. The extent of conversion for various initial pressures has been followed by gas chromatography by allowing the reaction to proceed for a time with various initial pressures:
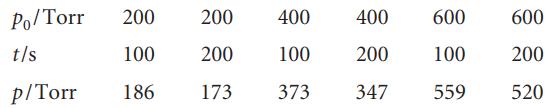
where p0 is the initial pressure and p is the final pressure of cyclopropane.
What are the order and rate constant for the reaction under these conditions?
Transcribed Image Text:
Po/Torr 200 200 t/s 100 200 p/Torr 186 173 400 100 373 400 200 347 600 600 100 200 559 520
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
R k Ax and d Adt Rate for 1st condition Po P t 014 fo...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The decomposition of iodoethane in the gas phase proceeds according to the following equation: C2H5I(g) C2H4(g) + HI(g) At 660. K, k = 7.2 10-4 s-1; at 720. K, k = 1.7 10-2 s-1. What is the rate...
-
The decomposition of acetaldehyde was studied in the gas phase at 791 K. The results of the measurements are shown as follows: Initial concentration (in mol/L) 9.72x10 -3 4.56x10 -3 Half-life (in s)...
-
In the gas phase at 500.C, cyclopropane reacts to form propene in a first-order reaction. The figure below shows the concentration of cyclopropane plotted versus time. Use the graph to calculate...
-
Plot the six risks on a probability/impact matrix using the project impact matrix 2023 template. Write one to two sentences stating the rationale for how you determined the quadrant placement for...
-
Simmons Market Research conducted a national consumer study of 13,787 respondents. A subset of the respondents was asked to indicate the primary source of the vitamins or mineral supplements they...
-
Background: SO MUCH CANDY DATA, SERIOUSLYCandy hierarchy data for 2017 Boing Boing Halloween candy hierarchy. This is survey data over the span of 4-years. The data is split into 4 separate files....
-
Africa offers a fast growing and exciting mobile phone market, with more than 50% growth per year since 2002. More than 28% of African consumers own mobile phones; a larger market than North America....
-
Assume that IBM leased equipment that was carried at a cost of $150,000 to Sharon Swander Company. The term of the lease is 6 years beginning January 1, 2011, with equal rental payments of $30,044 at...
-
The local theater company is choosing its next play. The company's members recently held a vote to decide which of three choices - Elektra, Tartuffe, and Hamlet - the company will perform. The...
-
Figure shows a uniform beam subject to a linearly increasing distributed load. The equation for the resulting elastic curve is w. (-X* + 21X - L*X) 120 EIL Based on the above equation determine the...
-
A second-order reaction of the type A + B P was carried out in a solution that was initially 0.050 mol dm 3 in A and 0.080 mol dm 3 in B. After 1.0 h the concentration of A had fallen to 0.020 mol...
-
The rate constant for the first-order decomposition of N 2 O 5 in the reaction 2 N 2 O 5 (g) 4 NO 2 (g) + O 2 (g) is k = 3.38 10 5 s 1 at 25C. What is the half-life of N 2 O 5 ? What will be the...
-
Charlotte Lee was named as a defendant partner in a lawsuit against a partnership. Her interest was as follows: Lee's husband had been a named partner in the business and when he died, his interest...
-
Founded in 1939 in a garage, Hewlett-Packard is the original Silicon Valley company. The HP Way is the legendary culture of innovation and creativity that the iconic company had codified and...
-
What are the advantages and disadvantages of "Passive Safety" in nuclear reactors?
-
Explain fundamental safety principles and the three- and five-levels safety of nuclear reactors.
-
How did the decision in Hedley Byrne v. Heller & Partners address the issue in Question 5?
-
We have a parliament to pass laws, a government to administer laws, and a police department to enforce laws. Ironically, these potent instruments for the restriction of liberty are necessary for the...
-
A pet food company has a business objective of expanding its product line beyond its current kidney- and shrimp-based cat foods. The company developed two new products, one based on chicken liver and...
-
Make an argument that Williams had a right to delay the closing until after August 1.
-
The rate constant for the decomposition of a certain substance is 1.70 x 10-2 dm3 mol-1 S-1 at 24C and 2.01 X 10-2 dm3 mol-1 S-1 at 37"C. Evaluate the Arrhenius parameters of the reaction.
-
Predict the order of magnitude of the isotope effect on the relative rates of displacement of (a) IH and 3H, (b) 160 and ISO. Will raising the temperature enhance the difference? Take kr(C-H) = 450 N...
-
The effective rate constant for a gaseous reaction that has a Lindemann-Hinshelwood mechanism is 1.7 X 10-3 s-I at 1.09 kPa and 2.2 X 10-4 S-1 at 25 Pa. Calculate the rate constant for the activation...
-
Imagine a wire with current I is directed at an angle 8, shown below. F = 17B) out of page Cross-sectional area A The wire is located within in a magnetic field. Using the coordinate system, we see...
-
You are still orbiting earth, as one of the astronauts on the International Space Station. In moving between compartments, you come to a complete stop and cannot reach any handholds. Describe several...
-
what is the final velocity if the mass is 0.150kg and the spring PE is 0.14625J and the initial kinetic energy is 0.14625J and spring compression is 0.05m and slope is -34.91?

Study smarter with the SolutionInn App


