Question: Evaluate the electronic partition function for atomic Fe at 298 K given the following energy levels. Level (1) Energy (cm) Degeneracy 415.9 704.0 888.1 3
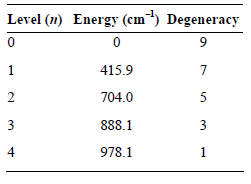
Level (1) Energy (cm) Degeneracy 415.9 704.0 888.1 3 978.1 2. 3. 4.
Step by Step Solution
3.50 Rating (173 Votes )
There are 3 Steps involved in it
q ge 9e 7eB4159 cm 0 1 SeB7040cm 4159 cm ... View full answer

Get step-by-step solutions from verified subject matter experts


