Question: How do the results shown in Figure 20.10 differ from the predictions of the Bohr model of the H atom? Figure 20.10 1s 2p 2s
Figure 20.10
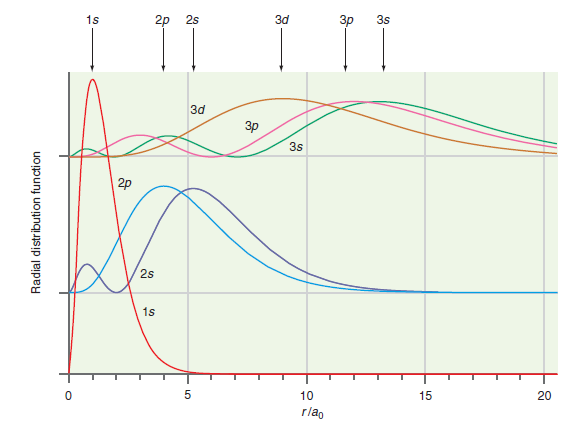
1s 2p 2s d 3s d 3s 2p 2s 1s 10 15 20 rlag Radial distribution function
Step by Step Solution
3.48 Rating (164 Votes )
There are 3 Steps involved in it
The Bohr model predicts that the electron ... View full answer

Get step-by-step solutions from verified subject matter experts


