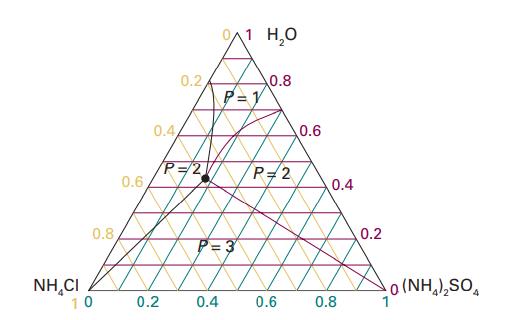
Referring to Fig. 5.8, deduce the molar solubility of (i) NH 4 Cl, (ii) (NH 4 )
Question:
Referring to Fig. 5.8, deduce the molar solubility of (i) NH4Cl, (ii) (NH4)2SO4 in water at 25 °C.
Data in Fig. 5.8,
Transcribed Image Text:
0.8 NHẠCI 10 0.6 0.2 0.44 0.2 P=2 0/1 H₂O P=1 fg P=3 0.4 0.8 P=2 0.6 0.6 0.4 0.8 0.2 0(NH,),SO.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (8 reviews)
Answer i The molar solubilit...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The molar solubility of MnCO3 is 4.2 3 1026 M. What is Ksp for this compound?
-
The diffusion coefficient of glucose in water at 25 C is 6.73 10 10 m 2 s 1 . Estimate the time required for a glucose molecule to undergo a root-mean-square displacement of 5.0mm.
-
Liquid water at 25 C and 1 bar fills a rigid vessel. If heat is added to the water until its temperature reaches 50 C, what pressure is developed? The average value of ( between 25 and 50oC is 36.2 (...
-
Mickey Limited is a manufacturing business that uses a standard costing system. The companys flexed budget for April 20X9 is: Sales 173,340 Costs Direct materials (22,500) Direct labour (37,684)...
-
Perlu Products, an IFRS reporter, reported an impairment loss of $ 65,000 for one of its plant assets on December 31, 2015. At December 31, 2016, the assets recoverable amount increased by $ 90,000....
-
Consider the following speedup of the insertion sort algorithm of Special Topic 14.2. For each element, use the enhanced binary search algorithm that yields the insertion position for missing...
-
Use the Hubble expansion relation (9.1.1), the temperature scaling relation (9.1.3), and the energy density relation before the electron-positron annihilation (9.3.6b) to show that the temperature as...
-
The chart of accounts of LR Company includes the following selected accounts. 112 Accounts Receivable ......401 Sales Revenue 120 Inventory ...........412 Sales Returns and Allowances 126 Supplies...
-
Consider that the current world price for copper ore it's $3.90 per pound. Suppose that domestic market for copper ore in Chile is described by the following demand and supply equation respectively P...
-
You have been asked to help prepare the financial statements of Orb, a limited liability company, for the year ended 31 December 2020. Orb's trial balance at 31 December 2020 is set out below. Orb -...
-
Use the phase diagram in Fig. 5.3 to state (i) the solubility of Ag in Sn at 800 C and (ii) the solubility of Ag 3 Sn in Ag at 460 C, (iii) the solubility of Ag 3 Sn in Ag at 300 C. Data in Fig. 5.3...
-
Air is a mixture with mass percentage composition 75.5 (N 2 ), 23.2 (O 2 ), 1.3 (Ar). Calculate the entropy of mixing when it is prepared from the pure (and perfect) gases.
-
In 2020, Noshy Corporation had accounting income of $234,000 and taxable income of $184,000. The difference is due to the use of different depreciation methods for tax and accounting purposes. The...
-
Conduct individual inquiry and research into the Roles and Influences of a current global business(es). Demonstrates evidence of extensive inquiry and research in relation to ONE role and TWO...
-
Provide a brief overview of the primary objectives of compilation, review, and preparation engagements under SSARS. Describe the task objectives that differentiate one SSARS engagement from another....
-
He personally loaned $150,000 to his wife on a demand promissory note without interest, in order to help her purchase some land adjacent to her retailing business which she operated as a sole...
-
Trifold Framework in Healthcare: Primary prevention includes educating and communicating a strong organizational culture that promotes respect and professionalism. Healthcare leaders must develop...
-
in April Pratt corporation made an advanced payment of $14,000 to a local company for advertising that would run during October. It is now October 31 and the advertising has run assuming original...
-
Consider the feedback system shown in Figure DP7.14. The process transfer function is marginally stable. The controller is the proportional-derivative (PD) controller Gc(s) = KP + KDs. (a) Determine...
-
B made an issue of 150,000 $1 ordinary shares at a premium of 20% the proceeds of which is received by cheque. What is the correct journal to record this? A. Bank Share capital Share premium B. Bank...
-
The Joule coefficient is defined by (T /V) U = (1/C V )[P T(P/T) V ]. Calculate the Joule coefficient for an ideal gas and for a van der Waals gas.
-
What is the physical origin of the pressure difference across a curved liquidgas interface?
-
Using the result of Equation (3.8), (P/T) V = / , express as a function of and V m for an ideal gas, and as a function of b, , and V m for a van der Waals gas.
-
Kae is an office manager at an insurance broker. Recently he has identified several breaches of data protection legislation by multiple members of his team. Kae must identify the unresolved issue...
-
Subject: Constitutional Law in Malaysia Question: Map various types of power given to the Yang Di-Pertuan Agong. State also the brief explanation to each type.?
-
Many companies have staggered or "classified" boards, where the directors are placed into different classes with terms that expire at different times. What are the advantages of having a staggered...

Study smarter with the SolutionInn App


