The degree of dissociation, , of CO 2 (g) into CO(g) and O 2 (g) at high
Question:
The degree of dissociation, α, of CO2(g) into CO(g) and O2(g) at high temperatures was found to vary with temperature as follows:
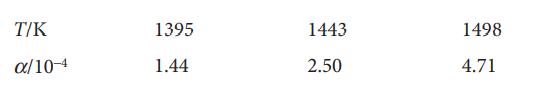
Assuming ΔrH⦵ to be constant over this temperature range, calculate K, ΔrG⦵, ΔrH⦵, and ΔrS⦵. Make any justifiable approximations.
Transcribed Image Text:
T/K α/10-4 1395 1.44 1443 2.50 1498 4.71
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
The equilibrium constant K for the reaction can be calculated using the following equation K 1 Using ...View the full answer

Answered By

Dansteve Matoke
As a consequence of more than three years of experience in ACADEMIC WRITING, I have vast, diverse knowledge and impeccable grammar. I guarantee the quality of my work across multiple fields. I intend to precisely meet the clients' expectations. Let's work together to achieve exceptional grades.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Estimate the degree of dissociation of a 0.200 m solution of nitrous acid (K a = 4.00 10 4 ) that is also 0.500 m in the strong electrolyte given in parts (a)(c). Use the data tables to obtain ,...
-
The degree of dissociation of HI at a particular temperature is 0.8. Calculate the volume (in lit) of 2M Na, S203 solution required to neutralise the iodine present in a equilibrium mixture of a...
-
The degree of dissociation of HI at a particular temperature of 0.8. Calculate the volume (in litre) of 1.6 M Na2 S,O3 solution required to neutralize the iodine present in an equilibrium mixture of...
-
Roberto finishes a triathlon (750-meter swim, 5-kilometer run, and 20-kilometer bicycle) in 63.2 minutes. Among all men in the race, the mean finishing time was 69.4 minutes with a standard deviation...
-
Fredrick Wilson Company determined that one of its finite- life intangible assets is impaired. The assets net carrying value on the date of the impairment is $ 905,000. Fredrick Wilson does not use a...
-
Use recursion to compute an, where n is a positive integer. Hint: If n is 1, then a n = a. If n is even, then an = (a n/2 ) 2 . Otherwise, a n = a a n1 .
-
Im gathering some information about the sales/collection process and how it is supposed to work. Okay?
-
The following information is available for two different types of businesses for the 2016 accounting year. Hopkins CPAs is a service business that provides accounting services to small businesses....
-
3. Consider a two period problem where a consumer has preferences over consumption in the two periods given by: log c + Blog c'. She has no initial assets and has income y in the first period y' in...
-
In a regression of average wages (W) on the number of employees (N) for a random sample of 30 firms, the following regression results were obtained a. How would you interpret the two regressions? b....
-
When the reaction A2 B advances by 0.10mol (that is, =+0.10mol) the Gibbs energy of the system changes by 6.4 kJmol 1 . What is the Gibbs energy of reaction at this stage of the reaction?
-
What is the justification for not including a pure liquid or solid in the expression for an equilibrium constant?
-
The U.S. Department of Transportation and the Federal Motor Carrier Safety Administration produce an annual report on various traffic violations. There were 2092 moving violations in the state of New...
-
List customer ID, customer name, order ID, order date, product ID and the quantity of each product ordered, for all customers who have placed orders. Hint: You need to include 3 tables in your FROM...
-
Select all that apply Materials activities include: (Check all that apply.) Multiple select question. factory overhead costs. purchasing materials. raw materials beginning inventory. selling finished...
-
Define Supply Chain Risk Management as the implementation of strategies to manage every day and exceptional risks.
-
What sort of data can be collected from an app such as MapMyRun? How can that be related to Under Armour's apparel division? In Cirque du Soleil, what data beyond an audience's facial expressions can...
-
Define the link between Leadership and Power. Give an example that explains the link.
-
Large antenna, as shown in Figure CP7.6(a), is used to receive satellite signals and must accurately track the satellite as it moves across the sky. The control system uses an armature-controlled...
-
Kims Konstructions has assembled the following data for a proposed straw-reinforced brick maker (SRBM): SRBM Cost: $26,000 Life: 5 years Revenue (p.a.) $11,000 Operating Expenses (p.a.) $3,000...
-
Determine the exchange current density from the information given in Exercise 25.16a. Data in Exercise 25.16a. The transfer coefficient of a certain electrode in contact with M 3+ and M 4+ in aqueous...
-
The transfer coefficient of a certain electrode in contact with M 3+ and M 4+ in aqueous solution at 25C is 0.39. The current density is found to be 55.0 mA cm 2 when the overvoltage is 125 mV. What...
-
The standard potentials of lead and tin are 126 mV and 136 mV respectively at 25C, and the overvoltage for their deposition are close to zero. What should their relative activities be in order to...
-
2. (4 pts) When leaning forward, the weight of the head (F) is supported by muscle tension in the neck (FM) and balanced by a force supplied by the upper vertebrae (F.). The weight is 50 N and the...
-
Briefly point out the important points of security of tenure for the judges in assuring judicial independence in Malaysia?
-
The same rocket sled drawn in the figure is decelerated at a rate of 196 m/s. What force (in N) is necessary to produce this deceleration? Assume that the rockets are off. The mass of the system is...

Study smarter with the SolutionInn App


