The relative permittivities of methanol (m.p. 95C) corrected for density variation are given below. What molecular information
Question:
The relative permittivities of methanol (m.p. −95°C) corrected for density variation are given below. What molecular information can be deduced from these values? Take ρ = 0.791 g cm−3 at 20°C.
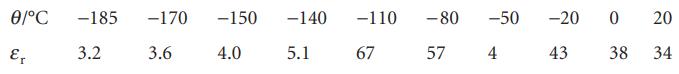
Transcribed Image Text:
Ꮎ/°C -185 Ꮛ 3.2 -170 -150 -140 3.6 4.0 5.1 -110 -80 67 57 -50 -20 4 43 0 38 20 34
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The two strongest absorption bands for methane occur at 3967 and 4...View the full answer

Answered By

Enock Oduor
I am a chemist by profession, i coach high school students with their homework, i also do more research during my free time, i attend educational and science fair seminars where i meet students and do some projects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Journalize the following transactions: a. Issued credit memo No. 2 to Rose, $50. b. Cash sales, $210 c. Received cheque from Tran Co., $90, less 3% discount. d. Bought merchandise on account from Mel...
-
The variation in the density of water, p , with temperature, T, in the range 20°C ¤ T ¤ 50°C, is given in the following table. Use these data to determine an empirical equation...
-
For following molecular model, where C atoms are black and H atoms are light blue: a. Write the molecular formula. b. Write the condensed structural formula. c. Write the IUPAC name.
-
The following information is available for Marigold's Hot Dogs: Actual production Budgeted production Standard direct labor hours Actual direct labor hours Standard variable overhead rate Actual...
-
Suppose a recent random sample of employees nationwide that have a 401(k) retirement plan found that 18% of them had borrowed against it in the last year. A random sample of 100 employees from a...
-
Hillside issues $4,000,000 of 6%, 15-year bonds dated January 1, 2018, that pay interest semiannually on June 30 and December 31. The bonds are issued at a price of $3,456,448. Required 1. Prepare...
-
Unless otherwise specified, which rule will always be processed?
-
Lexi Belcher picked up the monthly report that Irvin Santamaria left on her desk. She smiled as her eyes went straight to the bottom line of the report and saw the favorable variance for operating...
-
Phelps competed at the Olympics. He won several gold medals and cash prizes, all worth $300,000. In addition, in the year that Phelps competed in the Olympics, he received several commercial...
-
Clayton Kershaw of the Los Angeles Dodgers is one of the premier pitchers in baseball. His most popular pitch is a four-seam fastball. The data in the next column represent the pitch speed (in miles...
-
Suppose an H 2 O molecule ( = 1.85 D) approaches an anion. What is the favourable orientation of the molecule? Calculate the electric field (in volts per metre) experienced by the anion when the...
-
Which of the following molecules may be polar: CIF 3 , O 3 , H 2 O 2 ?
-
Explain when the export firm should no longer be concerned with the pricing of its products.
-
What is the X-11 model? What is it used for? Briefly explain how the X-11 model is used in forecasting.
-
Why was product analytics influential in Brand24s overall pricing optimization success?
-
What is the reason for differences in initial impact calculation versus the realized one (over $40k MRR vs. $4k MRR)?
-
What key considerations are required to develop a final commercial structure for B2B digital solutions?
-
Are businesses thinking about their services, about how they will play in the Web 3.0 world? For example, Facebook is transitioning to Meta.
-
Determine the utilization for each of these situations: a. A gas station has three pumps with an effective capacity to serve 20 customers per hour. Average demand is 45 customers per hour. b. An...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
The centre of the EPR spectrum of atomic deuterium lies at 330.02 mT in a spectrometer operating at 9.2482 GHz. What is the g-value of the electron in the atom?
-
A radical containing three equivalent protons shows a four-line spectrum with an intensity distribution 1:3:3:1. The lines occur at 331.4 mT, 333.6 mT, 335.8 mT, and 338.0 mT. What is the hyperfine...
-
A radical containing three in equivalent protons with hyperfine constants 2.11 mT, 2.87 m'T and 2.89 mT gives a spectrum centred on 332.8 mT. At what fields do the hyperfine lines occur and what are...
-
You ordered a Model DPS-8 Sony Blu-ray player from Orion Inc. on December 5 as a gift. Orion promised shipment by December 20. The Model DPS-8 Sony Blu-ray player didn't arrive until December 27,...
-
At the end of 2019 , Mcknight decides to adjust the manufacturing overhead rate to 170 % of direct labor cost. She explains to the company president that, because overhead was underallocated in 2019...
-
Company A uses the indirect method to prepare the statement of cash flows. The following select information was extracted from the company's records: Gain on sale of long term assets $4,000...

Study smarter with the SolutionInn App


