Use the phase diagram in Fig. 6.40 to state (a) The solubility of Ag in Sn at
Question:
Use the phase diagram in Fig. 6.40 to state
(a) The solubility of Ag in Sn at 800°C and
(b) The solubility of Ag3Sn in Ag at 460°C,
(c) The solubility of Ag3Sn in Ag at 300°C.
Fig. 6.40
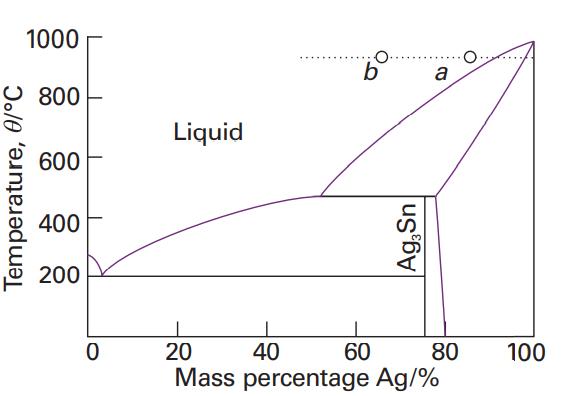
Transcribed Image Text:
Temperature, 0/°C 1000 800 600 400 200 0 Liquid O. b Ag,Sn 60 a 20 40 80 Mass percentage Ag/% 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Solution a The solubility of Ag in Sn at 800C is given by x...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the phase diagram in Fig. 6.41 to state (a) The solubility of B in A at 500C and (b) The solubility of AB2 in A at 390C, (c) The solubility of ABz in Bat 300e.
-
The phase diagram for neon is Temperature (K) Use the phase diagram to answer the following questions. (a) What is the approximate value of the normal melting point? (b) Over what pressure range will...
-
Use the phase diagram in Fig. 5.3 to state (i) the solubility of Ag in Sn at 800 C and (ii) the solubility of Ag 3 Sn in Ag at 460 C, (iii) the solubility of Ag 3 Sn in Ag at 300 C. Data in Fig. 5.3...
-
Passenger table (passengerid, address etc.) Flight table (flight id, departure, destination, depDate) Booking table (cID, fid, date, cost) a. Find Passengers who live in Chicago b. Total number of...
-
What is marking-to-market for a future? Why is this marking-to-market important for reducing counterparty risk?
-
As a means of enhancing heat transfer from high-performance logic chips, it is common to attach a heat sink to the chip surface in order to increase the surface area available for convection heat...
-
Air is stored in a tank where the pressure is \(40 \mathrm{psia}\) and the temperature is \(500{ }^{\circ} \mathrm{R}\). A converging-diverging nozzle with an exitto-throat area ratio of 2.5 attaches...
-
Wilken's Sandwich Shop maintains three separate menus for breakfast, lunch, and dinner. Gross margin computations for the three menu lines for 2012 are as follows:
-
Crane Company has several processing departments. Costs to be accounted for in the Assembly Department for November 2022 totaled $2,268,894 as follows. Work in process, November 1 Materials $78,800...
-
This is a new manufacturing corporation that issued $50000 common stock for cash on the first day. All overhead expenses are paid immediately by cash. To make it easy, there are no other operating...
-
Show that two phases are in mechanical equilibrium only if their pressures are equal.
-
Indicate on the phase diagram in Fig. 6.41 the feature that denotes incongruent melting. What is the composition of the eutectic mixture and at what temperature does it melt? Fig. 6.41 Temperature,...
-
Given a set P of n points, design an efficient algorithm for constructing a simple polygon whose vertices are the points of P.
-
H~3&500 1 2 4 6 Required: Determine the amount of goodwill impairment, if any, that should be recognized at December 31, 20X4, if the fair value of the Plastic reporting 8 unit is determined to be 9...
-
A comparison of the marketing methods of two multinational hotel brands: Marriot (hotels and resorts) and Premier Inn the ways in which these organisations market their products and services . This...
-
please plot graph when answering the questionUse the supply and demand analysis of the market for reserves to visually illustrate and explain how the following scenarios may arise in equilibrium....
-
What are some of the key considerations that organizations should keep in mind when selecting an ERP system?How do organizations typically approach the implementation of an ERP system, and what are...
-
Say for instance your in fashion marketing making a fashion marketing plan on Bareskin Candles & Co.'s Its mission statement is to provide customers with the glow and aroma to nourish their soul and...
-
A set of n elements in an n-dimensional linear space is a basis if and only if it is linearly independent.
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
How is the method of combination differences used in rotation vibration spectroscopy to determine rotational constants?
-
The wavenumber of the J=32 rotational transition of 1 H 35 Cl considered as a rigid rotor is 63.56 cm 1 ; what is the HCl bond length?
-
Classify the following rotors: (i) O 3 , (ii) CH 3 CH 3 , (iii) XeO 4 , (iv) FeCp 2 (Cp denotes the cyclopentadienyl group, C 5 H 5 ).
-
You have been analyzing your company's performance over the past few years and decided to conduct a horizontal analysis. 1. Some of the results are causing you concern. What are these and why do they...
-
Selma operates a contractor's supply store. She maintains her books using the cash method. At the end of 2023, her accountant computes her accrual basis income that is used on her tax return. For...
-
Schumacher Industries Inc. manufactures recreational vehicles. Schumacher Industries uses a job order cost system. The time tickets from June jobs are summarized as follows: Job 11-101 Job 11-102...

Study smarter with the SolutionInn App


