Question: Why does the list of elements for the D 6h group in Table 27.2 not list the elements C 2 6 , C 3 6
Why does the list of elements for the D6hgroup in Table 27.2 not list the elements C26, C36, and C46?
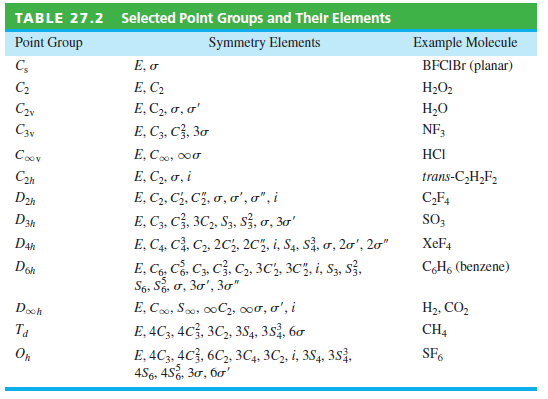
Selected Point Groups and Their Elements Symmetry Elements TABLE 27.2 Example Molecule BFCIB (planar) Point Group E, o E, C2 E, C2, o, o' E, C3, C3, 30 C, H2O2 C2 H,0 NF3 C2v C3y HCI E, C, 000 Coov trans-C,H,F, E, C2, o, i C,F4 E, C2, C3, C5, o, o', o", i Dah SO, E, C3, C, 3C,, S3, s, , 3' E, C4, C, C2, 2C3, 2C, i, S4, S, o, 2o', 20" E, C6, C, C3, C, C2, 3C2, 3C5, i, S3, s, S6, So, o, 30', 3" E, C0, So0, 00C2, 000, o', i E, 4C3, 4C3, 3C, 3S4, 3.S2, 60 D3h XEF4 Dah C,H, (benzene) Don H2, CO, Dooh CH4 Ta SF6 E, 4C3, 4C3, 6C,, 3C4, 3C,, i, 3S4, 383, , 452, 30, 6
Step by Step Solution
3.32 Rating (161 Votes )
There are 3 Steps involved in it
C 2 6 is not listed because it ... View full answer

Get step-by-step solutions from verified subject matter experts


