Which of the three normal modes of H 2 O in Figure 27.9 is best described as
Question:
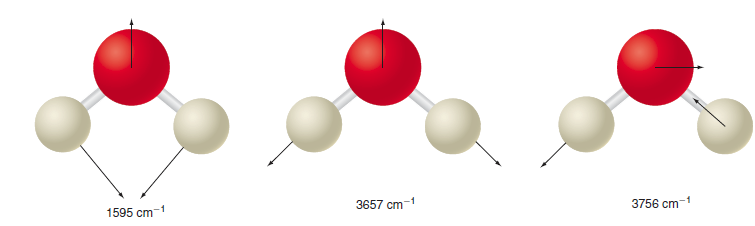
Transcribed Image Text:
3756 cm-1 3657 cm-1 1595 cm-1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The 1595 cm 1 mode does not involve significant changes in bo...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The organic molecules shown here are derivatives of benzene in which six-member rings are "fused" at the edges of the hexagons. (a) Determine the empirical formula of benzene and of these three...
-
Consider the molecule CH3Cl. (a) To what point group does the molecule belong? (b) How many normal modes of vibration does the molecule have? (c) What are the symmetries of the normal modes of...
-
Calculating the motion of individual atoms in the vibrational modes of molecules (called normal modes) is an advanced topic. Given the normal modes shown in the following figure, decide which of the...
-
help me pls P acquired \( 80 \% \) of the capital of S on 1 January 2021. At the year end 31 December 2021 the two companies have the following statements of financial position: Notes 1....
-
Solve the exponential equation algebraically. Approximate the result to three decimal places. 1. e4x = ex2+3 2. e3x = 25 3. 2x 3 = 29 4. e2x 6ex + 8 = 0
-
Trans Phone Repairs began operations on 1 December 2024 and completed the following transactions during the first month. 1. Jeremy Tran deposited \($15\)000 of his personal funds in a current account...
-
This exercise considers data from the Medical Expenditure Panel Survey (MEPS) described in Exercise 1.1 and Section 11.4. Our dependent variable consists of the number of outpatient (COUNTOP) visits....
-
In a recent year, Coach, Inc, a designer and marketer of handbags and other accessories, issued 12,100 shares of its $0.01 par value stock for $344,000 (these numbers are rounded). These additional...
-
Manufacturing costs for Davenport Company during 2018 were as follows: Beginning Finished Goods, January 1 Beginning Raw Materials, January 1 Beginning Work in Process, January 1 Direct Labor for the...
-
Consider the following pep9 trap: DECI number,d What 'd' refers in the above instruction? a) direct mode b) read(or input) decimal number c) read(or input) integer d) print( or output) decimal number
-
Can a molecule with an inversion center have a dipole moment? Give an example of a molecule with this symmetry element and explain your reasoning.
-
Why does the list of elements for the D 6h group in Table 27.2 not list the elements C 2 6 , C 3 6 , and C 4 6 ? Selected Point Groups and Their Elements Symmetry Elements TABLE 27.2 Example Molecule...
-
What are transferred in costs?
-
Shirley Swift transferred property with an adjusted basis of \(\$ 24,000\) and a fair market value of \(\$ 18,000\) to Alex Corporation in exchange for 100 shares of its Section 1244 stock. Three...
-
Find the osmotic pressure of a solution of 0.5 g of sucrose (C12H22O11) in 500 g water at 25C and 1 bar.
-
You have been assigned to perform tests of controls on the sales system at EDB Ltd. as part of the December 31, 2023, audit. EDB is a wholesaler of bathroom supplies such as vanity units, toilets,...
-
Consider two semiconductors, one made of Si and the other made of Ge. Both are at room temperature and doped with 10 7 phosphorous atoms. (a) Which semiconductor has the greater number of free...
-
Assume that the real risk-free rate is r* = 2% and the average expected inflation rate is 3% for each future year. The DRP and LP for Bond X are each 1%, and the applicable MRP is 2%. What is Bond Xs...
-
In problem, solve each logarithmic equation. Express irrational solutions in exact form and as a decimal rounded to three decimal places. 2 log 5 x = 3 log 5 4
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
For a black body, the temperature and the wavelength of emission maximum, max , are related by Wiens law, max T = 1/5c 2 , where c 2 =hc/k (see Problem 7A.3). Values of max from a small pinhole in...
-
Describe the properties of wave packets in terms of the Heisenberg uncertainty principle.
-
A glow-worm of mass 5.0 g emits red light (650nm) with a power of 0.10W entirely in the backward direction. To what speed will it have accelerated after 10 y if released into free space and assumed...
-
Explain why leasing is an option for a company expansion. include, what leasing is and how it will benefit the company in it's expanding efforts. Also, how is capital or operating leasing recorded on...
-
Discuss the following statement: " A head of state signs a treaty on behalf of his country in excess of authority of his country, such treaty shal be void for inconsistency with domestic law of the...
-
A company is looking at new equipment with an installed cost of $415,329. This cost will be depreciated straight-line to zero over the project's 5-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


