Question: Match the function with its graph using horizontal asymptotes as an aid. [The graphs are labeled (a), (b), (c), (d), (e), and (f).] (a) (b)
Match the function with its graph using horizontal asymptotes as an aid. [The graphs are labeled (a), (b), (c), (d), (e), and (f).]
(a)
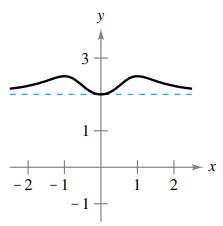
(b)
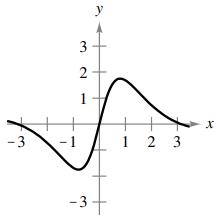
(c)
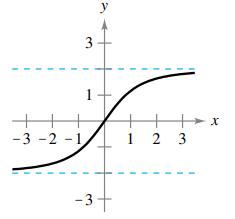
(d)
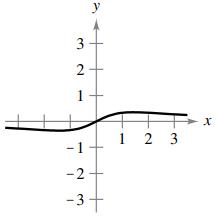
(e)
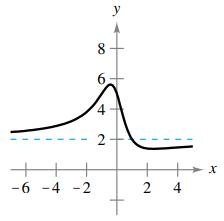
(f)
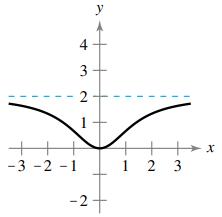
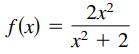
-2 -1 3 1 y 1 2 X
Step by Step Solution
3.43 Rating (159 Votes )
There are 3 Steps involved in it
option f is correc... View full answer

Get step-by-step solutions from verified subject matter experts


