Question: Sulfur hexafluoride (SF6) is a high-density gas that has refractive index, ns = 1.8 at a specified pressure, temperature, and wavelength. Consider the retro-reflecting prism
Sulfur hexafluoride (SF6) is a high-density gas that has refractive index, ns = 1.8 at a specified pressure, temperature, and wavelength. Consider the retro-reflecting prism shown in Fig. 12.16, that is immersed in SF6. Light enters through a quarter-wave antireflective coating and then totally reflects from the back surfaces of the glass. In principle, the beam should experience zero loss at the design wavelength (Pout= Pin).
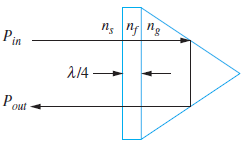
(a) Determine the minimum required value of the glass refractive index, ng, so that the interior beam will totally reflect.
(b) Knowing ng, find the required refractive index of the quarter-wave film, nf.
(c) With the SF6 gas evacuated from the chamber, and with the glass and film values as previously found, find the ratio, Pout/Pin. Assume very slight misalignment, so that the long beam path through the prism is not retraced by reflected waves.
, | Pin 214 out
Step by Step Solution
3.49 Rating (169 Votes )
There are 3 Steps involved in it
a We set the critical angle of total reflection equal to 45 which give... View full answer

Get step-by-step solutions from verified subject matter experts


