a. Use Figure 25-31 to select a tetrahydrofuran/water tetrahydrofuran/water mobile phase strength equivalent to the strength of
Question:
a. Use Figure 25-31 to select a tetrahydrofuran/water tetrahydrofuran/water mobile phase strength equivalent to the strength of 80% 80% methanol.
b. Describe how to prepare 1 liter of this tetrahydrofuran mobile phase.
c. What limitations would be imposed by the use of tetrahydrofuran?
Figure 25-31
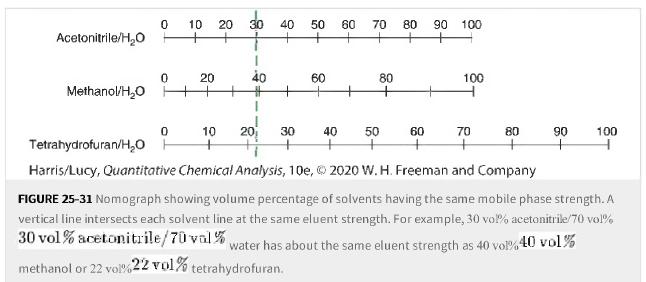
Transcribed Image Text:
Acetonitrile/H₂O 0 10 20 0 Methanol/H₂O + 0 Tetrahydrofuran/H₂O H 20 methanol or 22 vol%4 30 40 40 + # + 1 10 20 30 50 + 60 60 70 80 90 60 80 + + Harris/Lucy, Quantitative Chemical Analysis, 10e, © 2020 W. H. Freeman and Company 80 + + 40 50 100 100 70 90 + 100 FIGURE 25-31 Nomograph showing volume percentage of solvents having the same mobile phase strength. A vertical line intersects each solvent line at the same eluent strength. For example, 30 vol% acetonitrile/70 vol% 30 vol% acetonitrile/70 val% water has about the same eluent strength as 40 vol%40 vol% 22 vol% tetrahydrofuran.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
Answer a To select a tetrahydrofuranwater mobile phase strength equival...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
How to prepare 1 liter of a buffer mixture of pH = 9.5 starting from HCl and 10 mmol of Na2CO3? What will the pH value be after adding to 100 ml of the buffer mixture: a) 5.10-2 mmol of NaOH? b)...
-
This figure shows phase 1 vapor recovery from an underground gasoline storage tank. Before phase 1 vapor recovery was developed, gasoline vapors were vented directly into the air. Now the vapor is...
-
The phase diagram for water is shown in FIGURE 17-37. (a) What is the temperature T1 on the phase diagram? (b) What is the temperature T2 on the phase diagram? (c) What happens to the...
-
Assume you are given the following abbreviated financial statements: On the basis of this information, calculate as many liquidity, activity, leverage, profitability, and common stock measures as you...
-
Use the data in Table 11.6 on page 707. Assume that we wish to fit a simple linear regression model for predicting logarithm of 1980 price from logarithm of 1970 price. a. Find the posterior...
-
For the molecules shown in 24.85, Data from 24.85 Identify each of the functional groups in these molecules: (a) (Responsible for the odor of cucumbers) (b) (Quinine an antimalarial drug) (c)...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Brett Dunlop is seeking part-time employment while he attends school. He is considering purchasing technical equipment that will enable him to start a small training services company that will offer...
-
Why is the relational model more important above the other models?
-
Construct a market-value balance sheet for FedEx, using the information in Table 7.1 and stock prices reported in Sections 7.1 and 7.2. Assume that market and book values are equal for current...
-
The figure shows reversed-phase retention data for three compounds. a. Identify whether compounds A, B, and C are weak acids or bases. For each compound, what is the pKap K a and the retention factor...
-
a. UHPLC can provide exquisite resolution when run on long columns at high pressure or rapid separations with reasonable resolution if short columns are run fast. The drug acetaminophen run on a...
-
True or false? If the demand function is x 1 = p 1 , then the inverse demand function is x = 1/p 1 .
-
Analyse the following cost data: Employee training in problem solving - $25,000 Scrap Considering only the quality costs in the above data, Can you compute all 4 different types of quality costs:...
-
Managers face risky decisions every day. One method of controlling risk in a decision is conducting market research. This effort is quite necessary for new product development. Many commercial...
-
Changes to the financing cost target are made because of different market pointers trying to gauge monetary patterns and in this manner keep the market on target towards accomplishing the...
-
The Burj Khalifa project is a multi-use development tower with a total floor area of 460,000 square meters that includes residential, hotel, commercial, office, entertainment, shopping, leisure, and...
-
Using this code only. How would I scan a .dat file for it to be the maze instead of manually putting the values of the maze on the code? Also, how would I print the maze also? The top of the .dat...
-
Reconsider Prob. 5-52E. Using EES (or other) software, investigate the effect of the rate of cooling of the compressor on the exit temperature of air. Let the cooling rate vary from 0 to 100 Btu/lbm....
-
A. Select a recent issue (paper or online) of Report on Business Magazine, Canadian Business Magazine (online only), Bloomberg Businessweek, Fast Company, The Economist, or another business magazine....
-
Express the molecular mass ( uncertainty) of C9H9O6N3 with the correct number of significant figures.
-
Express the molecular mass ( uncertainty) of C9H9O6N3 with the correct number of significant figures.
-
Express the molecular mass ( uncertainty) of C9H9O6N3 with the correct number of significant figures.
-
How would you/how does your organization assess a contract to ensure that you can meet the requirements of a contract? How do you evaluate a contract prior to bidding on it? By leveraging external...
-
Identify someone who you consider as an excellent leader.What type of leadership style(s)does he/she exhibit? What conflict management toolcana leader use to diffuse conflict(s) in an organization?...
-
1. Speak to the determination of a profit or loss for a company (5 marks) 2. Differentiate between logistics costs and supply chain costs (5 marks) 3. Explain how does logistics costs impact supply...

Study smarter with the SolutionInn App


