Consider the cyclic voltammogram of the Co 3+ compound Co(B 9 C 2 H 11 )2. Suggest
Question:
Consider the cyclic voltammogram of the Co3+ compound Co(B9C2H11)2̅. Suggest a chemical reaction to account for each wave. Are the reactions reversible? How many electrons are involved in each step? Sketch the sampled current and square wave polarograms expected for this compound.
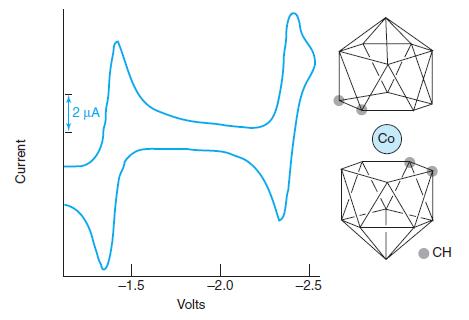
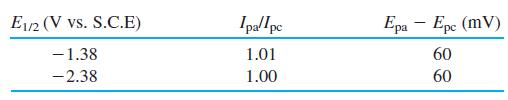
Transcribed Image Text:
2 µA Co CH -1.5 -2.0 -2.5 Volts Current
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
A cyclic voltammogram of the Co3 compound CoB9C2H112 provides information about the electrochemical ...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Consider the following energy profile (a) How many elementary reactions are in the reaction mechanism? (b) How many intermediates are formed in the reaction? (c) Which step is rate limiting? (d) Is...
-
For each of the following chemical reactions, write the correct element and/or compound symbols, formulas, and coefficients needed to produce complete, balanced equations. In all cases, the reactants...
-
A catalyst affects which one or more of the following? a. the energy needed for a chemical reaction to occur b. the energy a chemical reaction gives off c. the speed of a chemical reaction d. whether...
-
Your geothermal heat pump is used to heat your house in the winter. The heat pump is operated as a vapor-compression cycle. The design is such that there must be a 10 degree temperature difference...
-
Last week's total number of hours worked by a student, y, depends on the number of days, x, he reported to work last week. Suppose the data from nine students provided (a) Plot the scatter diagram....
-
Refer to the 2017 financial statements and related disclosure notes of FedEx Corporation. The financial statements can be found at the companys website (www.fedex.com). Required: 1. Does FedEx...
-
Describe the staffing process in an MNE. How does it differ from that of a domestic-only corporation?
-
1. How should Oreos success in social engagement be evaluated? What metrics would be most useful? Consider both the usage-specific metrics that a social media provider might supply as well as...
-
1 . differentiate between data elements and data types 2 . distinguish among different types of modern databases 3 . explain how businesses identify risks and opportunities associated with emerging...
-
Suppose General Motors (GM) stock sells for 25 dollars a share today. The stock will pay a dividend of 2 dollars at the end of the year. The annual risk free rate is 4 percent. (a) What is the...
-
A type of instruction that produce several lines of machine language code is a (A) Assemble (B) Address (C) Mnemonic (D) Macro
-
In a coulometric Karl Fischer water analysis, 25.00 mL of pure dry methanol required 4.23 C to generate enough I 2 to react with the residual H 2 O in the methanol. A suspension of 0.847 6 g of...
-
Draw the two isomers of bromopropane, C 3 H 7 Br.
-
Design a Social Welfare Function with specific merit goods where weights are assigned to individual-based preferences versus community-based preferences. Is there any practical example of a function...
-
In HTTP version 1.0, a server marked the end of a transfer by closing the connection. Explain why, in terms of the TCP layer, this was a problem for servers. Find out how HTTP version 1.1 avoids...
-
If a lake becomes used for bathing thanks to a remediation project, how do you value the benefit? Is it correct to consider the use-value associated with it and leave out the non-use value?
-
Estimate the probabilities of finding two messages with the same MD5 checksum, given total numbers of messages of 2 63 , 2 64 , and 2 65 . This is the birthday problem and again the probability that...
-
Show the result of the moves on Rubik's cube indicated in Problems 3-29. Remember that R, F, L, B, T, and U mean rotate \(90^{\circ}\) clockwise the right, front, left, back, top, and under faces,...
-
The September 30, 2017, balance sheet of Nelson Products reported the following: Accounts Receivable ........................................... $310,000 Allowance for Doubtful Accounts (credit...
-
a. What is meant by the term tax haven? b. What are the desired characteristics for a country if it expects to be used as a tax haven? c. What are the advantages leading an MNE to use a tax haven...
-
Using the linear calibration curve in Figure 4-13, find the quantity of unknown protein that gives a measured absorbance of 0.264 when a blank has an absorbance of 0.095. Figure 4-13 0.40 Unknown...
-
Consider the least-squares problem in Figure 4-11. (a) Suppose that a single new measurement produces a y value of 2.58. Find the corresponding x value and its uncertainty. (b) Suppose you measure y...
-
Consider the linear calibration curve in Figure 4-13, which is derived from the 14 corrected absorbances in the shaded region at the right side of Table 4-7. Create a least-squares spreadsheet like...
-
In Boston, due to the large amount of snowfall during winter of 2014-2015, the city has brought in several "snow dragons" to melt the snow. Model SND5400 is rated at: 180 ton snow rated capacity per...
-
How does the three-tier model effectively meet the literacy development needs of individual students? How does the response to intervention model integrate the three-tiers of instruction? What are...
-
TCS was established in 1968 with its headquarters in Mumbai. It was formed as a division of Tata Sons Limited (TSL), one of India's largest business conglomerates, and was called 'Tata Computer...

Study smarter with the SolutionInn App


