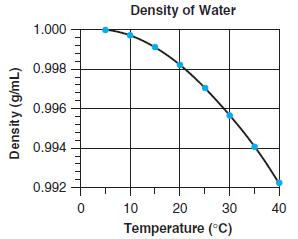
Reproduce the spreadsheet in Figure 2-23 and the graph in Figure 2-24. Figure 2-24 Density of Water
Question:
Reproduce the spreadsheet in Figure 2-23 and the graph in Figure 2-24.
Figure 2-24
Transcribed Image Text:
Density of Water 1.000 0.998 0.996 - 0.994 0.992 10 20 30 40 Temperature (°C) Density (g/mL)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
1 DensitygmL Temperature C 2 3 4 5 6 7 8 9 1...View the full answer

Answered By

NOEL BABUEF UNDI
I am mechanical engineer from an affiliated University in India called Rajiv Gandhi University of Knowledge and Technology. I have enough experience in Python programming and Microsoft Excel. I am very enthusiastic in mathematical problem solving. And worked as part time tutor for juniors in my university.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
Density helps us predict whether something will float. Density is super important to consider when building things like ships and submarines. The experiment in the video examines the water density by comparing a glass of water containing sugar with a simple glass of water
Students also viewed these Engineering questions
-
Reproduce FIGURE Q22.13 on your paper. Then draw a dot (or dots) on the figure to show the position (or positions) where an electron would experience no net force. | | | FIGURE Q22.13
-
Reproduce FIGURE Q23.2 on your paper. For each part, draw a dot or dots on the figure to show any position or positions (other than infinity) where E = 0. a. b. +'+
-
Reproduce FIGURE Q25.12 on your paper. Then draw a dot (or dots) on the figure to show the position (or positions) at which the electric potential is zero. | FIGURE Q25.12
-
Alert Security Services Co. offers security services to business clients. The trial balance for Alert Security Services Co. has been prepared on the following end-of-period spreadsheet for the year...
-
When might the statute of limitations be longer than the general statute you described in problem 11?
-
Prove that the set of edges picked in line 4 of APPROX-VERTEX-COVER forms a maximal matching in the graph G.
-
What are the criteria used for estimating the total effort required by a salesperson to cover a territory?
-
Jackson Company is a manufacturer of rocking chairs. Each chair passes through the assembly department and finishing department. This problem focuses on the assembly department. Direct materials are...
-
1. List and explain what benefits Mimitos will obtain with the creation of a website and online sales platform.
-
Forecasting with the Parsimonious Method and Estimating Share Value Using the ROPI Model Following are income statements and balance sheets for Cisco Systems. Cisco Systems Consolidated Statements of...
-
Water was drained from a buret between the 0.12- and 15.78-mL marks. The apparent volume delivered was 15.78 - 0.12 =15.66 mL. Measured in the air at 22C, the mass of water delivered was 15.569 g....
-
Reproduce the spreadsheet in Figure 2-23 and the graph in Figure 2-24. Figure 2-23 Figure 2-24 A E F G H Calculating Density of H2O with Equation 2-4 (from the delightful book by Dan Harris) 3...
-
R-134a at 30oC, 800 kPa is throttled so it becomes cold at 10oC. What is exit P?
-
Find the 69 th term of the sequence \[s_{n}=7-3 n \]
-
Classify the financial problems in Problems 16-19, and then answer each question by assuming a \(12 \%\) interest rate compounded annually. Find the value of a \(\$ 1,000\) certificate in 3 years.
-
Use a calculator to evaluate the present value of an annuity formula \[P=m\left[\frac{1-\left(1+\frac{r}{n}ight)^{-n t}}{\frac{r}{n}}ight]\] for the values of the variables \(m, r\), and \(t\)...
-
Find the 15 th term of the sequence \(s_{n}=4 n-3\).
-
What do we mean by a lump-sum problem?
-
Modify the employee scheduling model so that employees are paid $10 per hour on weekdays and $15 per hour on weekends. Change the objective so that you now minimize the weekly payroll. (You can...
-
What is the difference between the straight-line method of depreciation and the written down value method? Which method is more appropriate for reporting earnings?
-
After safety features and safety procedures in your laboratory have been explained to you, make a list of them.
-
Why is the buoyancy correction equal to 1 in Figure 2-9 when the density of the object being weighed is 8.0 g/mL? Figure 2-9
-
Pentane (C 5 H 12 ) is a liquid with a density of 0.626 g/mL near 25C. Find the true mass of pentane when the mass in air is 14.82 g. Assume air density = 0.001 2 g/mL.
-
Develop a SWOT analysis of Coles potential business. b. Can Cole be profitable and if so, in what period of time? c. What are the major business assumptions that require verification? d. Does Cole,...
-
The current temperature is 12 oC and the dew point is -29 oC. What is the relative humidity in percentage? Answer to one decimal place. The current temperature is 12 oC and the dew point is -29 oC....
-
The density of the block depends on what material the block is made out of. The table below shows the average densities for 3 different kinds of Earth materials. Using these data, calculate the...

Study smarter with the SolutionInn App


