Question: Why is the buoyancy correction equal to 1 in Figure 2-9 when the density of the object being weighed is 8.0 g/mL? Figure 2-9 1,003
Why is the buoyancy correction equal to 1 in Figure 2-9 when the density of the object being weighed is 8.0 g/mL?
Figure 2-9
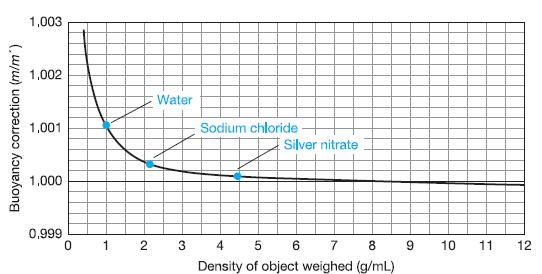
1,003 1,002 Water 1,001 Sodium chloride E Silver nitrate 1,000 0,999 1 4 5 6 7 8 10 11 12 Density of object weighed (g/mL) Buoyancy correction (m/m")
Step by Step Solution
3.32 Rating (149 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


