Question: Table 19-1 gives examples of standard methods for analyses using a spectrophotometric reaction. a. What is a standard method of analysis? b. U.S. Environmental Protection
Table 19-1 gives examples of standard methods for analyses using a spectrophotometric reaction.
a. What is a standard method of analysis?
b. U.S. Environmental Protection Agency method 350.1 for ammonia requires that a blank sample spiked with a known amount of analyte be analyzed within each batch of samples. What is the purpose of the spiked blank sample?
c. Standard Method 4500-Cl E for chloride states that particulate matter should be removed by filtration or centrifugation. Why must particulate be removed?
Table 19-1
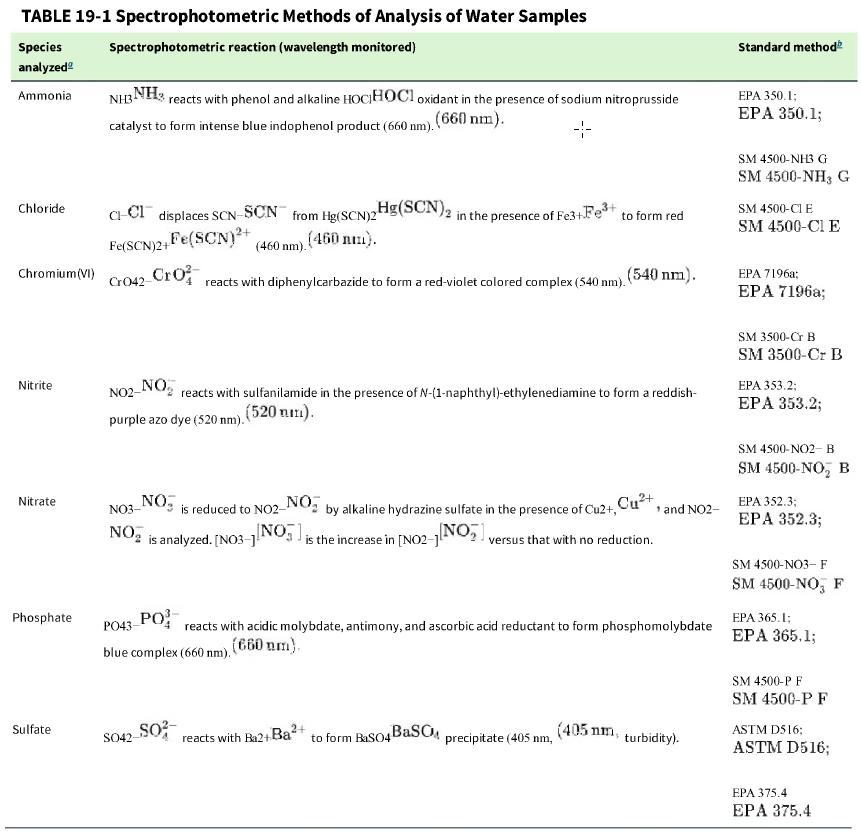
TABLE 19-1 Spectrophotometric Species analyzed Ammonia Chloride Chromium (VI) Nitrite Nitrate Phosphate Sulfate Spectrophotometric reaction (wavelength monitored) NH3NH3 reacts with phenol and alkaline HOCHOCI oxidant in the presence of sodium nitroprusside catalyst to form intense blue indophenol product (660 nm). (660) nm). 4- Methods of Analysis of Water Samples C-Cl displaces SCN-SCN from Hg(SCN)2 Fe(SCN)2+ Fe(SCN)+ (460 nm). (450 nm). Cro42-Cro NO2- NO purple azo dye (520 nm). reacts with diphenylcarbazide to form a red-violet colored complex (540 nm). Hg(SCN) 2 in the presence of Fe3+Fe+ to form red NO3- NO is analyzed. [NO3-] SO42-SO reacts with sulfanilamide in the presence of N-(1-naphthyl)-ethylenediamine to form a reddish- (520 mm). NO, NO is reduced to NO2- by alkaline hydrazine sulfate in the presence of Cu2+, Cu+, and NO2- [NO]; is the increase in [NO2-1[NO versus that with no reduction. PO43- PO blue complex (660 nm). (660 nm). (540 nm). reacts with acidic molybdate, antimony, and ascorbic acid reductant to form phosphomolybdate 2+ reacts with Ba2+ Ba to form BaSO4BaSOM precipitate (405 nm, (405 nm, turbidity). Standard method EPA 350.1; EPA 350.1; SM 4500-NH3 G SM 4500-NH3 G SM 4500-CI E SM 4500-CI E EPA 7196a; EPA 7196a; SM 3500-Cr B SM 3500-Cr B EPA 353.2; EPA 353.2; SM 4500-NO2-B SM 4500-NO, B EPA 352.3; EPA 352.3; SM 4500-NO3- F SM 4500-NO, F EPA 365.1; EPA 365.1; SM 4500-P F SM 4500-P F ASTM D516: ASTM D516; EPA 375.4 EPA 375.4
Step by Step Solution
3.63 Rating (157 Votes )
There are 3 Steps involved in it
Answer a A standard method of analysis is a specific set of steps that must be followed ... View full answer

Get step-by-step solutions from verified subject matter experts


