(a) Butler-volmer equation and redox system question The Butler-Volmer equation for a one electron transfer is given...
Question:
(a) Butler-volmer equation and redox system question
The Butler-Volmer equation for a one electron transfer is given by:
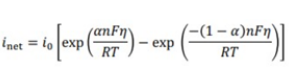
When the over potential is so small that F?/RT
![]() The exchange current density of a electrode at 298 K is 0.79 mA cm -2 . using equation 1 , assume the overpotential is +5.0 mV and RT/F = 25.7 mV at 298K and:
The exchange current density of a electrode at 298 K is 0.79 mA cm -2 . using equation 1 , assume the overpotential is +5.0 mV and RT/F = 25.7 mV at 298K and:
(i) Calculate the current density
(ii) Calculate the current going through an electrode Of a total area Of 5 cm 2
(b) Consider a hypothetical one-electron redox system, M 2+ M 3+ . Assume that [M 2+ l > [M 3+ ] and that the rate of diffusion is the same for M 2+ and M 3+
(i) Draw a labeled sketch plot of the current density versus overpotential, clearly labeling the oxidation/reduction regions and the charge transfer and mass transfer control regions. (ii) Show how the plot under (i) is expected to change if the temperature increases.





