Question: The following thermochemical equation is for the reaction of CO(g) with NO(g) to form CO(g) and N (9). 2CO(g) + 2NO(g) 2CO2(g) + N(g)
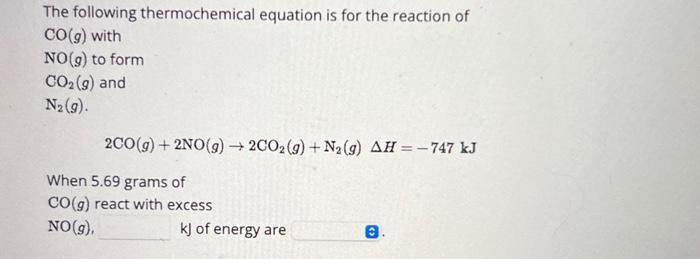
The following thermochemical equation is for the reaction of CO(g) with NO(g) to form CO(g) and N (9). 2CO(g) + 2NO(g) 2CO2(g) + N(g) AH = -747 kJ When 5.69 grams of CO(g) react with excess NO(g), kJ of energy are 0.
Step by Step Solution
3.44 Rating (160 Votes )
There are 3 Steps involved in it
Solutions To determine when 5699 1 first its Motor Calculate Moles of the mass amount ... View full answer

Get step-by-step solutions from verified subject matter experts


