In a certain chemical plant, a reversible fluid-phase isomerization A?B is carried out over a solid catalyst
Question:
In a certain chemical plant, a reversible fluid-phase isomerization A?B is carried out over a solid catalyst in a tubular packed-bed reactor. If the reaction is so rapid that mass transfer between the catalyst surface and the bulk fluid is rate-limiting, show that the kinetics are described in terms of the bulk concentrations C A and C B by
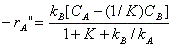
where -r " A = moles of A reacting per unit area catalyst per time k A , k B = mass transfer coefficients for A and B K = reaction equilibrium constant
It is desired to double the capacity of the existing plant by processing twice the feed of reactant A while maintaining the same fractional conversion of A to B in the reactor. How much larger a reactor, in terms of catalyst weight, would be required if all other operating variables are held constant? You may use the Thoenes-Kramaers correlation for mass transfer coefficients in a packed bed. Describe the effects of the flow rate, temperature, and particle size on conversion.

Introduction to Operations Research
ISBN: 978-1259162985
10th edition
Authors: Frederick S. Hillier, Gerald J. Lieberman




