Suppose you start with a 500 ml beaker of water that contains two drops (~0.l ml) of
Fantastic news! We've Found the answer you've been seeking!
Question:
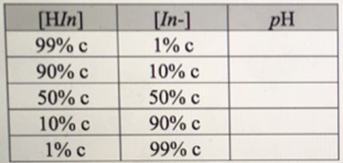
Suppose you start with a 500 ml beaker of water that contains two drops (~0.l ml) of an indicator dye with a known pK a . Let the total concentration of the indicator dye, [Hln] + [112?], be equal to c. By adding a few drops of concentrated acid (or base) to this solution, you are able to shift the pH of the solution significantly. Fill in the following data for this solution. You should express your pH values in terms of the pK a of the indicator.
Related Book For 

Posted Date:




