Question: (1) Aryl azide is an excellent photo-crosslinker (reacting to form covalent bonds with other molecules when activated by ultraviolet light) to label protein of
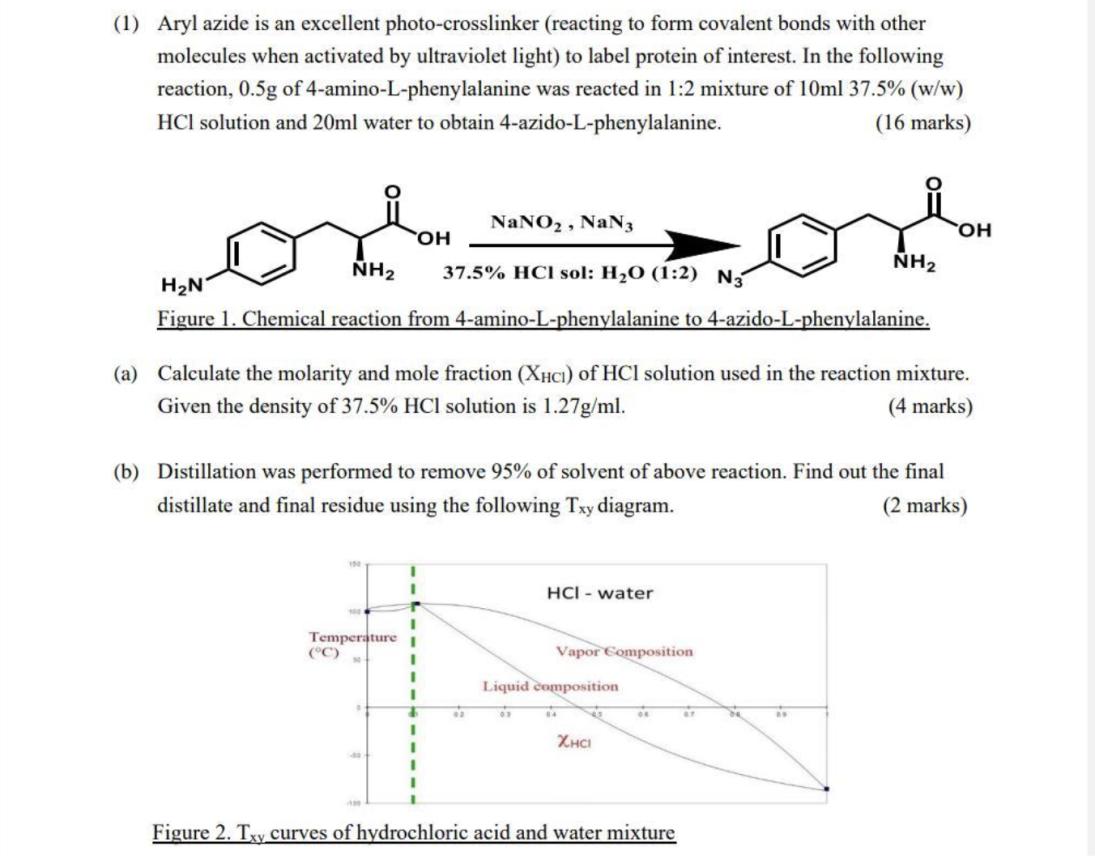

(1) Aryl azide is an excellent photo-crosslinker (reacting to form covalent bonds with other molecules when activated by ultraviolet light) to label protein of interest. In the following reaction, 0.5g of 4-amino-L-phenylalanine was reacted in 1:2 mixture of 10ml 37.5% (w/w) HCl solution and 20ml water to obtain 4-azido-L-phenylalanine. (16 marks) OH NH HN Figure 1. Chemical reaction from 4-amino-L-phenylalanine to 4-azido-L-phenylalanine. I Temperature I (C) I . 4 1 I NaNO, NaN3 (a) Calculate the molarity and mole fraction (XHCI) of HCI solution used in the reaction mixture. Given the density of 37.5% HCl solution is 1.27g/ml. (4 marks) (b) Distillation was performed to remove 95% of solvent of above reaction. Find out the final distillate and final residue using the following Txy diagram. (2 marks) I 4 I I 1 1 37.5% HCI sol: HO (1:2) N3 42 sor 65 HCI- water Liquid composition Vapor Composition 14 XHCI NH 04 Figure 2. Txy curves of hydrochloric acid and water mixture OH ar (A Txy diagram/curve represents a binary mixture, the compositions in this diagram are expressed as mole fraction of HCl.) (c) After distillation, 95% of the solvent was removed, 100ml of acetonitrile (ACN, a common laboratory used intermediate polarity, aprotic solvent, water miscible solvent) was added to the reaction mixture. State the Observation and explain why it happen. (5 marks) (d) It is known that higher acidity speeds up the reaction from aryl amine to aryl azide, 25ml of 37.5% (w/w) HCl was used instead of 10 ml in part (a), determine the final distillate and final residue again to remove 95% of solvent in above reaction using the Txy diagram from part (b). (5 marks)
Step by Step Solution
3.56 Rating (160 Votes )
There are 3 Steps involved in it
answer a Sure heres how to calculate the molarity and mole fraction of HCl solution used in the reac... View full answer

Get step-by-step solutions from verified subject matter experts


