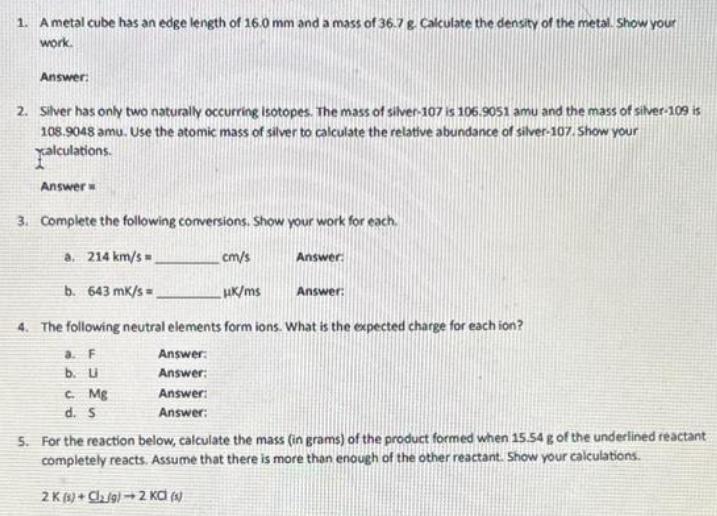
1. A metal cube has an edge length of 16.0 mm and a mass of 36.7...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
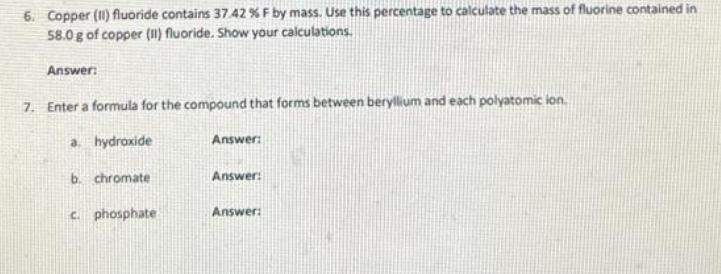
1. A metal cube has an edge length of 16.0 mm and a mass of 36.7 g. Calculate the density of the metal. Show your work. Answer: 2. Silver has only two naturally occurring isotopes. The mass of silver-107 is 106.9051 amu and the mass of silver-109 is 108.9048 amu. Use the atomic mass of silver to calculate the relative abundance of silver-107. Show your xalculations. Answer 3. Complete the following conversions. Show your work for each. a. 214 km/s b. 643 mk/s cm/s Answer HK/ms Answer: 4. The following neutral elements form ions. What is the expected charge for each ion? a. F Answer: b. U Answer: C. Mg Answer: d. S Answer: 5. For the reaction below, calculate the mass (in grams) of the product formed when 15.54 g of the underlined reactant completely reacts. Assume that there is more than enough of the other reactant. Show your calculations. 2K (1) Cl₂ g) 2 KC (s) 6. Copper (1) fluoride contains 37.42% F by mass. Use this percentage to calculate the mass of fluorine contained in 58.0 g of copper (1) fluoride. Show your calculations. Answer: 7. Enter a formula for the compound that forms between beryllium and each polyatomic ion. a. hydroxide Answer: b. chromate Answer: c. phosphate Answer: 1. A metal cube has an edge length of 16.0 mm and a mass of 36.7 g. Calculate the density of the metal. Show your work. Answer: 2. Silver has only two naturally occurring isotopes. The mass of silver-107 is 106.9051 amu and the mass of silver-109 is 108.9048 amu. Use the atomic mass of silver to calculate the relative abundance of silver-107. Show your xalculations. Answer 3. Complete the following conversions. Show your work for each. a. 214 km/s b. 643 mk/s cm/s Answer HK/ms Answer: 4. The following neutral elements form ions. What is the expected charge for each ion? a. F Answer: b. U Answer: C. Mg Answer: d. S Answer: 5. For the reaction below, calculate the mass (in grams) of the product formed when 15.54 g of the underlined reactant completely reacts. Assume that there is more than enough of the other reactant. Show your calculations. 2K (1) Cl₂ g) 2 KC (s) 6. Copper (1) fluoride contains 37.42% F by mass. Use this percentage to calculate the mass of fluorine contained in 58.0 g of copper (1) fluoride. Show your calculations. Answer: 7. Enter a formula for the compound that forms between beryllium and each polyatomic ion. a. hydroxide Answer: b. chromate Answer: c. phosphate Answer:
Expert Answer:
Answer rating: 100% (QA)
1 A metal cube has an edge of 160mm and a mass of 367g Calculate the density of the metal Solution 1 Identify the data and what to find Edge length of the cube s 160 mm convert to meters later Mass of ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Miller-Rabin test to check whether a number N is composite. This will involve computing a N1 mod N for some value of a. [10 marks] Carry out the steps for N = 65 and a = 1, 2, 8 and 12. on what each...
-
In this lab you will write a class called Time. The Time class will have three data members: hours, minutes, and seconds (all ints). Times will be stored using a 24 hour clock, so 10 AM is stored as...
-
Chiz enterprise reports its gross sales/receipts during the quarter as follows: Sale of computers Gross receipts - computer repairs Sales returns and allowances Sales discounts for early payments...
-
What are the implications of treating losses as passive?
-
An exchange rate is 0.7000 and the six-month domestic and foreign risk-free interest rates are 5% and 7% (both expressed with continuous compounding). What is the six-month forward rate? Need to know...
-
The company is committed to ethical conduct and has no tolerance for fraud and unethical behavior. There are some concerns about abuses in your department. Do you know anything about the concerns I...
-
The following events took place for Chaterjee Manufacturing Company during January 2014, the first month of its operations as a producer of digital thermometers: a. Purchased $ 95,200 of materials....
-
Schultz Incorporated ("Schultz") is a California manufacturer. Schultz employs several hourly, nonexempt workers in assembly-line positions. As a result, they are entitled to receive meal and rest...
-
Add parenthesis to the expression 6 * 1 - 2 to change its value from 4 to -6.
-
Explain why an increase in the rate at which a company uses a particular part increases the optimal order size of that part, while an increase in carrying costs decreases the optimal order size.
-
Again with Moviedata.xlsx, for each movie, extract the number of copies purchased from this data. You probably want to use the SUBSTITUTE function. The syntax of the SUBSTITUTE function is...
-
Two firms, Secure Inc. and Gamble Corp. are identical except that they have different capital structures. Both companies expect to generate a net operating profit of $100,000 per year in perpetuity....
-
Salem Instruments sold $475,000 worth of 120-day commercial paper for $468,000. What is the dollar amount of interest paid on the commercial paper? What is the effective 120-day rate on the paper?
-
Assume that you are the CFO of a company contemplating a stock repurchase next quarter. Right now you forecast that the companys EPS will be $2.09, but you receive a large bonus if EPS exceeds $2.10....
-
Hingston Mansingh, a manufacturing company, has done exceptionally well with productivity, efficiency and percent value-added time ratios being consistently over 80% for the previous three years....
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
About half of the hydrochloric acid produced annually in the United States (3.0 billion pounds) is used in metal pickling. This process involves the removal of metal oxide layers from metal surfaces...
-
A sample of zinc metal reacts completely with an excess of hydrochloric acid: Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) The hydrogen gas produced is collected over water at 25.0C using an arrangement...
-
Three apprentice tailors (X, Y, and Z) are assigned the task of measuring the seam of a pair of trousers. Each one makes three measurements. The results in inches are X (31.5, 31.6, 31.4); Y (32.8,...
-
W hat is diauxic growth? Explain the roles of cAMP and CAP in this process.
-
What is antisense RNA? How does it affect the translation of a complementary mRNA?
-
List and describe three general ways that the functions of transcription factors can be modulated.

Study smarter with the SolutionInn App


