Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol)...
Fantastic news! We've Found the answer you've been seeking!
Question:
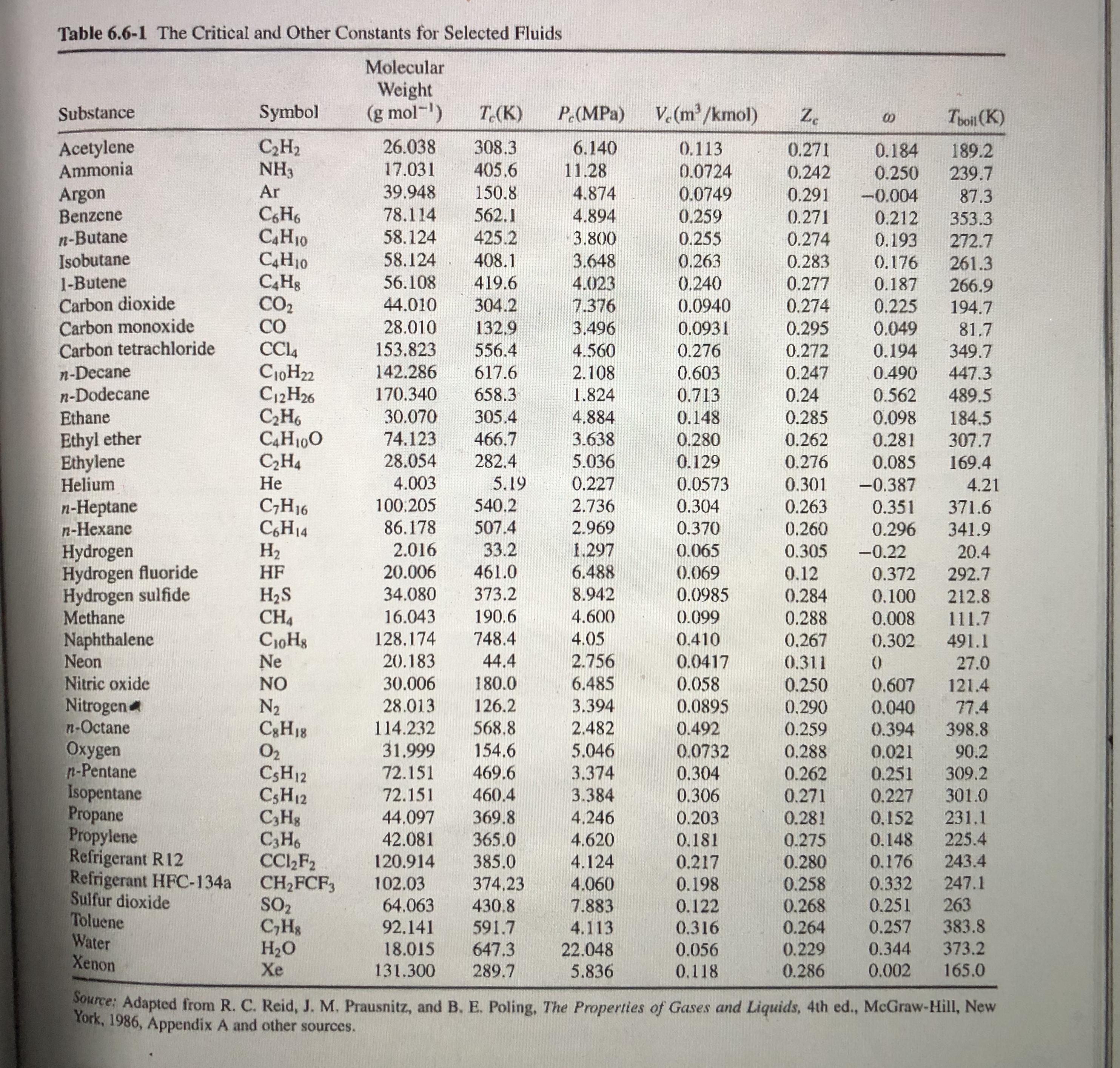

Transcribed Image Text:
Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140 0.113 0.271 0.184 0.250 189.2 17.031 11.28 0.0724 0.0749 0.242 239.7 Ar 39.948 150.8 Argon Benzene n-Butane Isobutane 1-Butene Carbon dioxide Carbon monoxide Carbon tetrachloride 4.874 0.291 -0.004 87.3 CH6 C4H10 C4H10 78,114 562.1 4.894 0.259 0.271 0.212 353.3 58.124 58.124 425.2 3.800 0.255 0.274 0.193 272.7 408.1 419.6 3.648 0.263 0.283 0.176 261.3 0.240 0.0940 56.108 4.023 0.277 0.187 266.9 194.7 CO2 CO 44.010 304.2 7.376 0.274 0.225 28.010 132,9 3.496 0.0931 0.295 0.049 81.7 349.7 153.823 556.4 4.560 0.276 0.272 0.194 n-Decane n-Dodecane Ethane C10H22 C12H26 C,H6 C4H100 C,H4 Не 142.286 617.6 2.108 0.603 0.247 0.490 447.3 170.340 658.3 1.824 0.713 0.24 0.562 489.5 30.070 305.4 4.884 0.148 0.285 0.098 184.5 307.7 Ethyl ether Ethylene Helium 74.123 466.7 3.638 5.036 0.280 0.262 0.281 28.054 282.4 0.129 0.276 0.085 169.4 4.003 5.19 0.227 0.0573 0.301 -0.387 4.21 540.2 n-Heptane n-Hexane C;H16 C,H14 H2 100.205 2.736 0.304 0.263 0.351 371.6 86.178 507.4 2.969 0.370 0.260 0.296 341.9 2.016 33.2 1.297 Hydrogen Hydrogen fluoride Hydrogen sulfide Methane Naphthalene Neon Nitric oxide Nitrogen n-Octane Oxygen n-Pentane Isopentane Propane Propylene Refrigerant R12 Refrigerant HFC-134a Sulfur dioxide Toluene Water Xenon 0.065 0.305 -0.22 20.4 | HF 20.006 461.0 6.488 (0.069 0.12 0.372 292.7 373.2 H2S CH4 CioHg Ne 34.080 8.942 0.0985 0.284 0.100 212.8 16.043 190.6 4.600 0.099 0.288 0.008 111.7 128,174 748.4 4.05 0.410 0.267 0.302 491.1 20.183 44.4 2.756 0.0417 0.058 0.0895 0.311 27.0 NO 30.006 180.0 6.485 0.250 0.607 121.4 N2 C3H18 O2 CSH12 CSH12 C,Hg C3H6 CC,F2 CH2FCF, SO2 C,Hs H2O Xe 28.013 126.2 3.394 0.290 0.040 77.4 114.232 568.8 2.482 0.492 0.259 0.288 0.394 398.8 31.999 154.6 5.046 0.0732 0.021 90.2 309.2 72.151 469.6 3.374 0.304 0.262 0.271 0.251 72.151 3.384 4,246 301.0 231.1 460.4 0.306 0.227 44.097 42.081 369.8 365.0 0.203 0.281 0,152 4.620 0.181 0.275 0.148 225.4 4.124 4.060 120.914 385.0 0.217 243.4 0.280 0.258 0.268 0.264 0.176 102.03 64.063 0.332 0.251 374,23 0.198 247.1 0.122 0.316 263 383.8 373.2 430.8 7.883 92.141 591.7 4.113 0.257 647.3 18.015 131.300 22.048 5.836 0.056 0.229 0.344 289.7 0.118 0.286 0.002 165.0 Source: Adapted from R. C. Reid, J. M. Prausnitz, and B. E. Poling, The Properties of Gases and Liquids, 4th ed., McGraw-Hill, New York, 1986, Appendix A and other sources. Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140 0.113 0.271 0.184 0.250 189.2 17.031 11.28 0.0724 0.0749 0.242 239.7 Ar 39.948 150.8 Argon Benzene n-Butane Isobutane 1-Butene Carbon dioxide Carbon monoxide Carbon tetrachloride 4.874 0.291 -0.004 87.3 CH6 C4H10 C4H10 78,114 562.1 4.894 0.259 0.271 0.212 353.3 58.124 58.124 425.2 3.800 0.255 0.274 0.193 272.7 408.1 419.6 3.648 0.263 0.283 0.176 261.3 0.240 0.0940 56.108 4.023 0.277 0.187 266.9 194.7 CO2 CO 44.010 304.2 7.376 0.274 0.225 28.010 132,9 3.496 0.0931 0.295 0.049 81.7 349.7 153.823 556.4 4.560 0.276 0.272 0.194 n-Decane n-Dodecane Ethane C10H22 C12H26 C,H6 C4H100 C,H4 Не 142.286 617.6 2.108 0.603 0.247 0.490 447.3 170.340 658.3 1.824 0.713 0.24 0.562 489.5 30.070 305.4 4.884 0.148 0.285 0.098 184.5 307.7 Ethyl ether Ethylene Helium 74.123 466.7 3.638 5.036 0.280 0.262 0.281 28.054 282.4 0.129 0.276 0.085 169.4 4.003 5.19 0.227 0.0573 0.301 -0.387 4.21 540.2 n-Heptane n-Hexane C;H16 C,H14 H2 100.205 2.736 0.304 0.263 0.351 371.6 86.178 507.4 2.969 0.370 0.260 0.296 341.9 2.016 33.2 1.297 Hydrogen Hydrogen fluoride Hydrogen sulfide Methane Naphthalene Neon Nitric oxide Nitrogen n-Octane Oxygen n-Pentane Isopentane Propane Propylene Refrigerant R12 Refrigerant HFC-134a Sulfur dioxide Toluene Water Xenon 0.065 0.305 -0.22 20.4 | HF 20.006 461.0 6.488 (0.069 0.12 0.372 292.7 373.2 H2S CH4 CioHg Ne 34.080 8.942 0.0985 0.284 0.100 212.8 16.043 190.6 4.600 0.099 0.288 0.008 111.7 128,174 748.4 4.05 0.410 0.267 0.302 491.1 20.183 44.4 2.756 0.0417 0.058 0.0895 0.311 27.0 NO 30.006 180.0 6.485 0.250 0.607 121.4 N2 C3H18 O2 CSH12 CSH12 C,Hg C3H6 CC,F2 CH2FCF, SO2 C,Hs H2O Xe 28.013 126.2 3.394 0.290 0.040 77.4 114.232 568.8 2.482 0.492 0.259 0.288 0.394 398.8 31.999 154.6 5.046 0.0732 0.021 90.2 309.2 72.151 469.6 3.374 0.304 0.262 0.271 0.251 72.151 3.384 4,246 301.0 231.1 460.4 0.306 0.227 44.097 42.081 369.8 365.0 0.203 0.281 0,152 4.620 0.181 0.275 0.148 225.4 4.124 4.060 120.914 385.0 0.217 243.4 0.280 0.258 0.268 0.264 0.176 102.03 64.063 0.332 0.251 374,23 0.198 247.1 0.122 0.316 263 383.8 373.2 430.8 7.883 92.141 591.7 4.113 0.257 647.3 18.015 131.300 22.048 5.836 0.056 0.229 0.344 289.7 0.118 0.286 0.002 165.0 Source: Adapted from R. C. Reid, J. M. Prausnitz, and B. E. Poling, The Properties of Gases and Liquids, 4th ed., McGraw-Hill, New York, 1986, Appendix A and other sources.
Expert Answer:

Related Book For 

Introduction to Statistical Quality Control
ISBN: 978-1118146811
7th edition
Authors: Douglas C Montgomery
Posted Date:
Students also viewed these chemical engineering questions
-
Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140...
-
Carbon dioxide gas enters a pipe at 3 MPa and 500 K at a rate of 2 kg/s. CO2 is cooled at constant pressure as it flows in the pipe and the temperature CO2 drops to 450 K at the exit. Determine the...
-
Carbon dioxide enters an adiabatic nozzle at 8 MPa and 450 K with a low velocity and leaves at 2 MPa and 350 K. Using the generalized enthalpy departure chart, determine the exit velocity of the...
-
3) Sauseda Corporation has two operating divisions-an Inland Division and a Coast Division. The company's Customer Service Department provides services to both divisions. The variable costs of the...
-
Discuss the convergence or the divergence of the series with nth term: (a) 2ne-n, (b) nne-n, (c) e-ln n, (d) (ln n)e-n, (e) n!e-n, (f) n!e-n2.
-
Which of the following statements best explains why distributors tend to spend less on storage space per square meter per year than retailers? a. Distributors provide break bulk service. b....
-
Piecewise Linear Regression. In Example 7.3 we showed how a linear regression model with a change in slope at some point $t\left(x_{\min } Example 7.3 An important special case of practical interest...
-
If five people from the same organization calculated manufacturing cycle efficiency for one specific process, would each compute the same MCE? Why or why not?
-
You have an outstanding student loan with required payments of $550 per month for the next four years. The interest rate on the loan is 11% APR (compounded monthly). Now that you realize your best...
-
Imagine that Howard has asked you to write some queries to help him make better use of his data. For each information request below, write a single query that provides the answer set. When a task...
-
You are just hired as a Working Capital Manager for a big manufacturing company. Your boss asked you to focus on inventory management right now. Explain how you are going to achieve your job tasks...
-
As noted on pages 355356, one key to being effective as a communicator is to be supportive of people. Supportive communication involves being accurate and honest with people in a manner that builds,...
-
Knowing how to discipline employees who behave inappropriately is an important managerial skill. The trick is to change the bad behavior into good behavior permanently, getting people to accept their...
-
A farm grows corn and produces eggs. The marginal cost of producing each of these products increases as more of it is produced. a. Make a graph that illustrates the farms PPF . b. The farm adopts a...
-
\(10 \mathrm{~kg}\) of wet steam of quality 0.8 at 5 bar pressure is heated at constant pressure till the temperature rise is \(500^{\circ} \mathrm{C}\). Calculate the amount of energy added as heat.
-
A container is filled with a saturated steam at 12 bar. The volume of the container is \(1 \mathrm{~m}^{3}\). First, the container is evacuated, then necessary amount of water is filled and...
-
Please explain which Excel formula you used, the reason behind your choice, and include Excel tables in the solution. An American baseball outfielder s $ 3 2 5 million contract with a baseball firm...
-
We all experience emotions, but some people disguise their true feelings better than others. Do you think this is a helpful or harmful thing to do? Under what conditions do you think it would be most...
-
The data in Table 9E.1 represent individual observations on molecular weight taken hourly from a chemical process. The target value of molecular weight is 1,050 and the process standard deviation is...
-
Consider the control chart shown in Exercise 5.17. Would the use of warning limits reveal any potential out-of-control conditions?
-
A supplier ships a product in lots of size N = 8,000. We wish to have an AOQL of 3%, and we are going to use single sampling. We do not know the suppliers process fallout but suspect that it is at...
-
Define a closely held corporation.
-
Explain the role played by a board of directors of a corporation.
-
Explain the steps in forming a limited liability corporation.

Study smarter with the SolutionInn App


