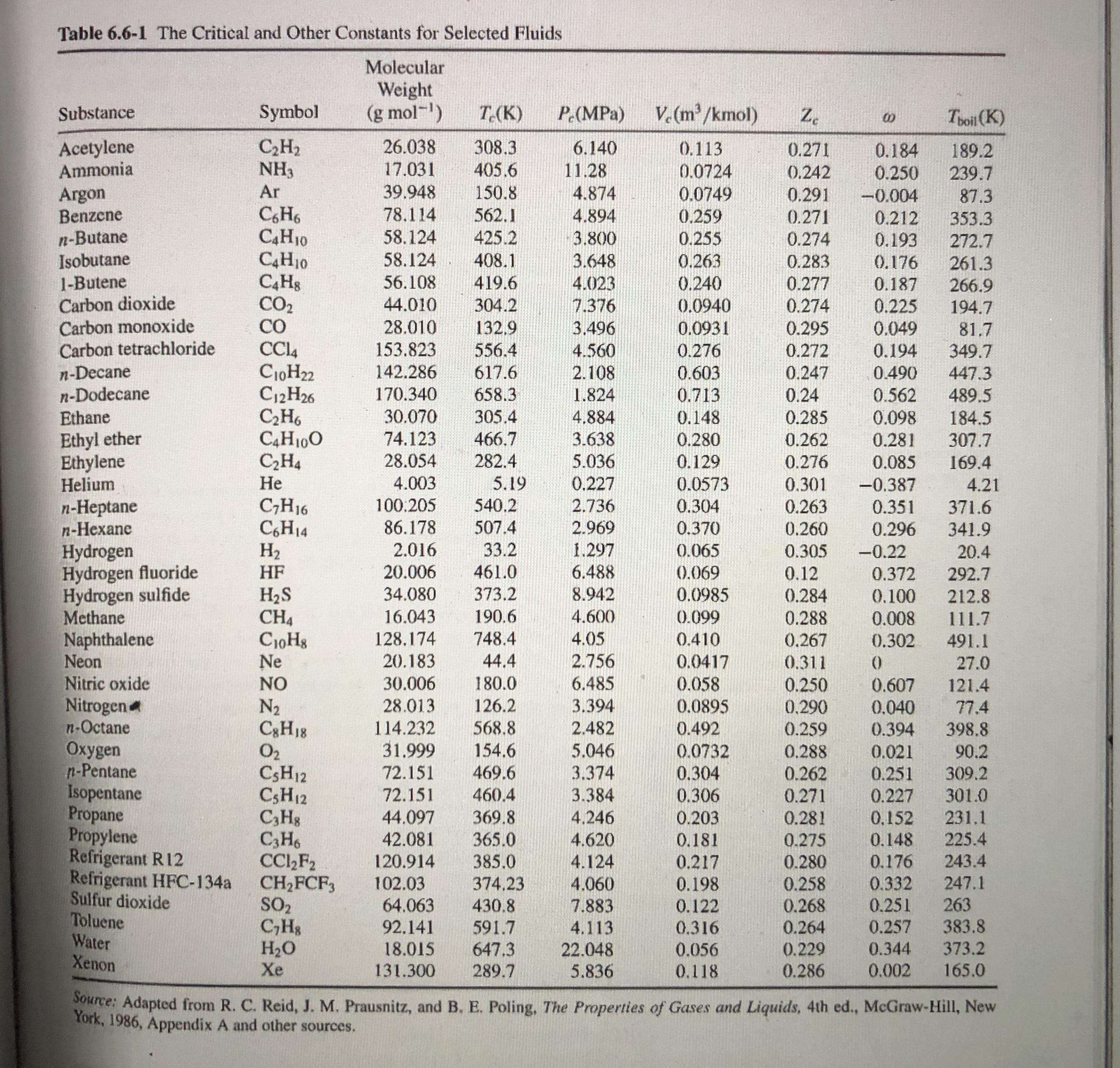
Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol)...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
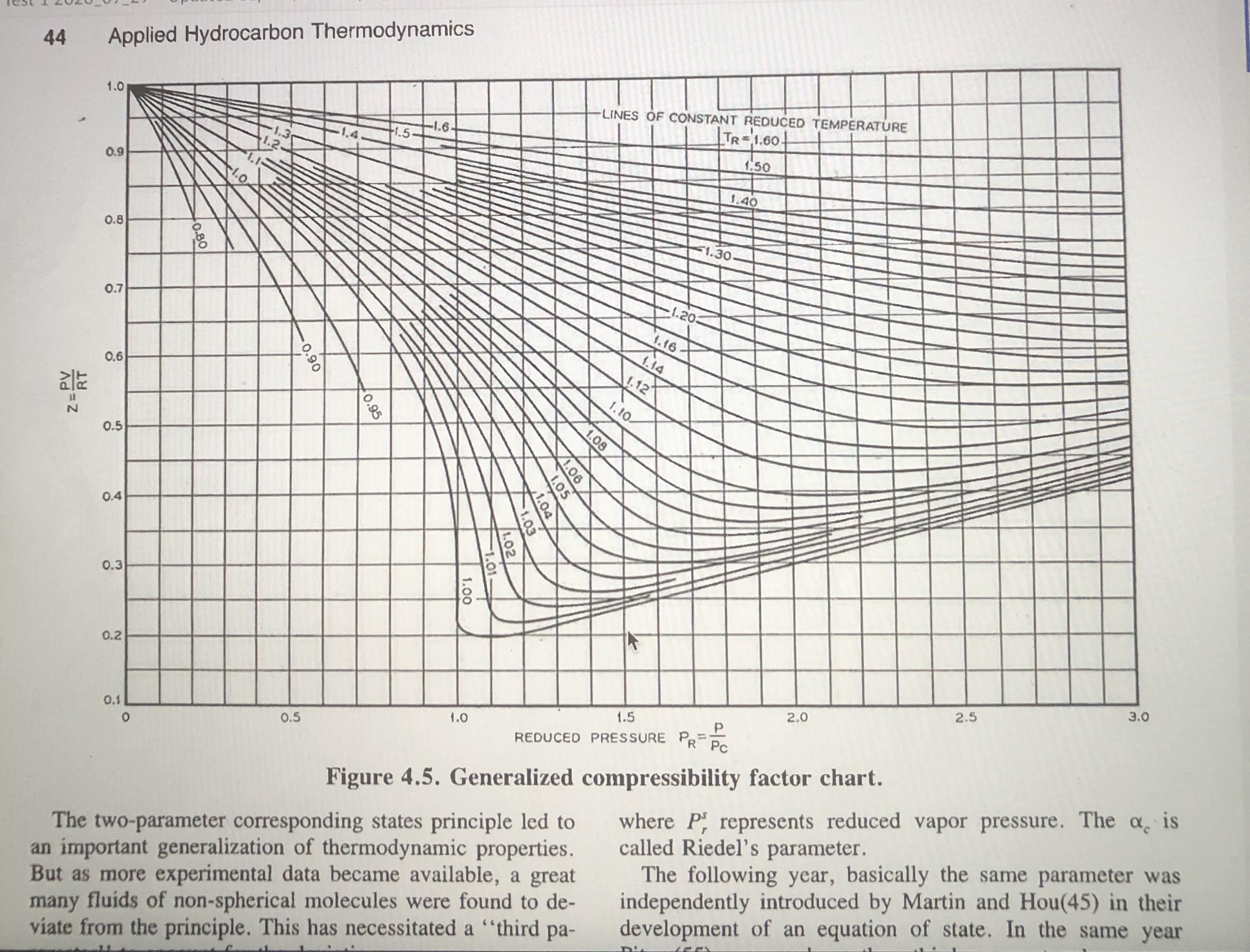
Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140 0.113 0.271 0.184 0.250 189.2 17.031 11.28 0.0724 0.0749 0.242 239.7 Ar 39.948 150.8 Argon Benzene n-Butane Isobutane 1-Butene Carbon dioxide Carbon monoxide Carbon tetrachloride 4.874 0.291 -0.004 87.3 CH6 C4H10 C4H10 78,114 562.1 4.894 0.259 0.271 0.212 353.3 58.124 58.124 425.2 3.800 0.255 0.274 0.193 272.7 408.1 419.6 3.648 0.263 0.283 0.176 261.3 0.240 0.0940 56.108 4.023 0.277 0.187 266.9 194.7 CO2 CO 44.010 304.2 7.376 0.274 0.225 28.010 132,9 3.496 0.0931 0.295 0.049 81.7 349.7 153.823 556.4 4.560 0.276 0.272 0.194 n-Decane n-Dodecane Ethane C10H22 C12H26 C,H6 C4H100 C,H4 Не 142.286 617.6 2.108 0.603 0.247 0.490 447.3 170.340 658.3 1.824 0.713 0.24 0.562 489.5 30.070 305.4 4.884 0.148 0.285 0.098 184.5 307.7 Ethyl ether Ethylene Helium 74.123 466.7 3.638 5.036 0.280 0.262 0.281 28.054 282.4 0.129 0.276 0.085 169.4 4.003 5.19 0.227 0.0573 0.301 -0.387 4.21 540.2 n-Heptane n-Hexane C;H16 C,H14 H2 100.205 2.736 0.304 0.263 0.351 371.6 86.178 507.4 2.969 0.370 0.260 0.296 341.9 2.016 33.2 1.297 Hydrogen Hydrogen fluoride Hydrogen sulfide Methane Naphthalene Neon Nitric oxide Nitrogen n-Octane Oxygen n-Pentane Isopentane Propane Propylene Refrigerant R12 Refrigerant HFC-134a Sulfur dioxide Toluene Water Xenon 0.065 0.305 -0.22 20.4 | HF 20.006 461.0 6.488 (0.069 0.12 0.372 292.7 373.2 H2S CH4 CioHg Ne 34.080 8.942 0.0985 0.284 0.100 212.8 16.043 190.6 4.600 0.099 0.288 0.008 111.7 128,174 748.4 4.05 0.410 0.267 0.302 491.1 20.183 44.4 2.756 0.0417 0.058 0.0895 0.311 27.0 NO 30.006 180.0 6.485 0.250 0.607 121.4 N2 C3H18 O2 CSH12 CSH12 C,Hg C3H6 CC,F2 CH2FCF, SO2 C,Hs H2O Xe 28.013 126.2 3.394 0.290 0.040 77.4 114.232 568.8 2.482 0.492 0.259 0.288 0.394 398.8 31.999 154.6 5.046 0.0732 0.021 90.2 309.2 72.151 469.6 3.374 0.304 0.262 0.271 0.251 72.151 3.384 4,246 301.0 231.1 460.4 0.306 0.227 44.097 42.081 369.8 365.0 0.203 0.281 0,152 4.620 0.181 0.275 0.148 225.4 4.124 4.060 120.914 385.0 0.217 243.4 0.280 0.258 0.268 0.264 0.176 102.03 64.063 0.332 0.251 374,23 0.198 247.1 0.122 0.316 263 383.8 373.2 430.8 7.883 92.141 591.7 4.113 0.257 647.3 18.015 131.300 22.048 5.836 0.056 0.229 0.344 289.7 0.118 0.286 0.002 165.0 Source: Adapted from R. C. Reid, J. M. Prausnitz, and B. E. Poling, The Properties of Gases and Liquids, 4th ed., McGraw-Hill, New York, 1986, Appendix A and other sources. 44 Applied Hydrocarbon Thermodynamics LINES OF CONSTANT REDUCED TEMPERATURE TR-1.60- 1.0 -1.6- 1.3 1.5 1.50 0.9 1.40 1.0 F1.30- 0.8 1.20 0.7 1.16- 1.14 1.12 0.6 1.10- 0.5 0.4 0.3 0.2 3.0 2.5 2.0 1.5 1.0 REDUCED PRESSURE P=. R Pc %3D 0.1 0.5 where P represents reduced vapor pressure. The a is called Riedel's parameter. The following year, basically the same parameter was independently introduced by Martin and Hou(45) in their development of an equation of state. In the same year Figure 4.5. Generalized compressibility factor chart. The two-parameter corresponding states principle led to an important generalization of thermodynamic properties. But as more experimental data became available, a great many fluids of non-spherical molecules were found to de- viate from the principle. This has necessitated a "third pa- 1.08 V1.06 1.05 0.95 0.90 1.00 0.80 RT Z=PV However, if the principle of corresponding states were exact, the vapor pressure curves for all the components, plotted in the reduced form, should result in one composite curve; that in practice does not occur due to differences in molecular structures of various substances. Therefore, to account for the deviation from the corresponding states principle, a third parameter called the acentric factor @ (see Reid et al.3 for listing of values for various pure components) is introduced in the generalized relationship expressed by Equation 11.3: Pr = exp(A+ @B) (11.4) %3D where A and B are expressed by the following equations: 6.09648 -1.28866 In(T, )+0.16934(T,)° T, A = 5.92714 – (11.5). 15.6875 B = 15.2518 – -13.4721 In(T;)+0.4357(T,)° (11.6) In Equation 11.4, since the product of A + @B_is dimensionless, vapor pressure P takes pertinent units that are used for critical pressure, P. The consistency of Equation 11.4 is evident because when T= T, consequently, T, = 1, the condition at which both A and B are 0 eventually resulting in the equality of P, and P, that is, at %3D V. Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140 0.113 0.271 0.184 0.250 189.2 17.031 11.28 0.0724 0.0749 0.242 239.7 Ar 39.948 150.8 Argon Benzene n-Butane Isobutane 1-Butene Carbon dioxide Carbon monoxide Carbon tetrachloride 4.874 0.291 -0.004 87.3 CH6 C4H10 C4H10 78,114 562.1 4.894 0.259 0.271 0.212 353.3 58.124 58.124 425.2 3.800 0.255 0.274 0.193 272.7 408.1 419.6 3.648 0.263 0.283 0.176 261.3 0.240 0.0940 56.108 4.023 0.277 0.187 266.9 194.7 CO2 CO 44.010 304.2 7.376 0.274 0.225 28.010 132,9 3.496 0.0931 0.295 0.049 81.7 349.7 153.823 556.4 4.560 0.276 0.272 0.194 n-Decane n-Dodecane Ethane C10H22 C12H26 C,H6 C4H100 C,H4 Не 142.286 617.6 2.108 0.603 0.247 0.490 447.3 170.340 658.3 1.824 0.713 0.24 0.562 489.5 30.070 305.4 4.884 0.148 0.285 0.098 184.5 307.7 Ethyl ether Ethylene Helium 74.123 466.7 3.638 5.036 0.280 0.262 0.281 28.054 282.4 0.129 0.276 0.085 169.4 4.003 5.19 0.227 0.0573 0.301 -0.387 4.21 540.2 n-Heptane n-Hexane C;H16 C,H14 H2 100.205 2.736 0.304 0.263 0.351 371.6 86.178 507.4 2.969 0.370 0.260 0.296 341.9 2.016 33.2 1.297 Hydrogen Hydrogen fluoride Hydrogen sulfide Methane Naphthalene Neon Nitric oxide Nitrogen n-Octane Oxygen n-Pentane Isopentane Propane Propylene Refrigerant R12 Refrigerant HFC-134a Sulfur dioxide Toluene Water Xenon 0.065 0.305 -0.22 20.4 | HF 20.006 461.0 6.488 (0.069 0.12 0.372 292.7 373.2 H2S CH4 CioHg Ne 34.080 8.942 0.0985 0.284 0.100 212.8 16.043 190.6 4.600 0.099 0.288 0.008 111.7 128,174 748.4 4.05 0.410 0.267 0.302 491.1 20.183 44.4 2.756 0.0417 0.058 0.0895 0.311 27.0 NO 30.006 180.0 6.485 0.250 0.607 121.4 N2 C3H18 O2 CSH12 CSH12 C,Hg C3H6 CC,F2 CH2FCF, SO2 C,Hs H2O Xe 28.013 126.2 3.394 0.290 0.040 77.4 114.232 568.8 2.482 0.492 0.259 0.288 0.394 398.8 31.999 154.6 5.046 0.0732 0.021 90.2 309.2 72.151 469.6 3.374 0.304 0.262 0.271 0.251 72.151 3.384 4,246 301.0 231.1 460.4 0.306 0.227 44.097 42.081 369.8 365.0 0.203 0.281 0,152 4.620 0.181 0.275 0.148 225.4 4.124 4.060 120.914 385.0 0.217 243.4 0.280 0.258 0.268 0.264 0.176 102.03 64.063 0.332 0.251 374,23 0.198 247.1 0.122 0.316 263 383.8 373.2 430.8 7.883 92.141 591.7 4.113 0.257 647.3 18.015 131.300 22.048 5.836 0.056 0.229 0.344 289.7 0.118 0.286 0.002 165.0 Source: Adapted from R. C. Reid, J. M. Prausnitz, and B. E. Poling, The Properties of Gases and Liquids, 4th ed., McGraw-Hill, New York, 1986, Appendix A and other sources. 44 Applied Hydrocarbon Thermodynamics LINES OF CONSTANT REDUCED TEMPERATURE TR-1.60- 1.0 -1.6- 1.3 1.5 1.50 0.9 1.40 1.0 F1.30- 0.8 1.20 0.7 1.16- 1.14 1.12 0.6 1.10- 0.5 0.4 0.3 0.2 3.0 2.5 2.0 1.5 1.0 REDUCED PRESSURE P=. R Pc %3D 0.1 0.5 where P represents reduced vapor pressure. The a is called Riedel's parameter. The following year, basically the same parameter was independently introduced by Martin and Hou(45) in their development of an equation of state. In the same year Figure 4.5. Generalized compressibility factor chart. The two-parameter corresponding states principle led to an important generalization of thermodynamic properties. But as more experimental data became available, a great many fluids of non-spherical molecules were found to de- viate from the principle. This has necessitated a "third pa- 1.08 V1.06 1.05 0.95 0.90 1.00 0.80 RT Z=PV However, if the principle of corresponding states were exact, the vapor pressure curves for all the components, plotted in the reduced form, should result in one composite curve; that in practice does not occur due to differences in molecular structures of various substances. Therefore, to account for the deviation from the corresponding states principle, a third parameter called the acentric factor @ (see Reid et al.3 for listing of values for various pure components) is introduced in the generalized relationship expressed by Equation 11.3: Pr = exp(A+ @B) (11.4) %3D where A and B are expressed by the following equations: 6.09648 -1.28866 In(T, )+0.16934(T,)° T, A = 5.92714 – (11.5). 15.6875 B = 15.2518 – -13.4721 In(T;)+0.4357(T,)° (11.6) In Equation 11.4, since the product of A + @B_is dimensionless, vapor pressure P takes pertinent units that are used for critical pressure, P. The consistency of Equation 11.4 is evident because when T= T, consequently, T, = 1, the condition at which both A and B are 0 eventually resulting in the equality of P, and P, that is, at %3D V.
Expert Answer:
Answer rating: 100% (QA)
Reduced Temperature and pressure In thermodynamics the reduced properties of a fluid are a set of state variables scaled by the fluids state propertie... View the full answer

Related Book For 

Introduction to Statistical Quality Control
ISBN: 978-1118146811
7th edition
Authors: Douglas C Montgomery
Posted Date:
Students also viewed these chemical engineering questions
-
Table 6.6-1 The Critical and Other Constants for Selected Fluids Molecular Weight (g mol-) V. (m/kmol) Substance Symbol T.(K) P (MPa) Z. Toil (K) Acetylene Ammonia C,H2 NH3 26.038 308.3 405.6 6.140...
-
Carbon dioxide gas enters a pipe at 3 MPa and 500 K at a rate of 2 kg/s. CO2 is cooled at constant pressure as it flows in the pipe and the temperature CO2 drops to 450 K at the exit. Determine the...
-
Carbon dioxide enters an adiabatic nozzle at 8 MPa and 450 K with a low velocity and leaves at 2 MPa and 350 K. Using the generalized enthalpy departure chart, determine the exit velocity of the...
-
Ronnie owns 600 shares of a stock mutual fund. This year he received dividend distributions of 60 stock mutual fund shares ($ 40 per share) and long term capital gain distributions of 45 stock mutual...
-
Let a > 0. Show that the series (1 + an)-1 is divergent if 0 < a < 1 and is convergent if a > 1.
-
A concrete pile 406 mm 406 mm in cross section is shown in Figure P12.13. Calculate the ultimate skin friction resistance by using the a. method [use Eq. (12.61) and Table 12.11] b. method c. ...
-
Consider the air pollution and mortality data given in Table B.15. a. Is there a problem with collinearity? Discuss how you arrived at this decision. b. Perform a ridge trace on these data. c. Select...
-
Eddy's Piano Rebuilding Company has been operating for one year (2010). At the start of 2011, its income statement accounts had zero balances and its balance sheet account balances were as follows:...
-
Show that the set {5, 15, 25, 35} is a group under multiplication modulo 40. What is the identity element of this group? Can you see any relationship between this group and U(8)? Additionally, A...
-
Bonds 1. Municipal Bonds - Municipal bonds are haircut per Exhibit 1 based on both their time to maturity and scheduled maturity at date of issue. 2. Corporate Bonds - Corporate bonds are haircut...
-
QUESTION 1 (25 marks) Meeting Corner (Pty.) Ltd. ("Meeting Corner") is a restaurant located in Stellenbosch, Western Cape. The entity's main clientele is employees from close-by businesses and...
-
Determine the magnitude of Earth's gravitational field for \((a)\) a 70. 0-kg person standing at Earth's surface, \((b)\) a \(700.0-\mathrm{kg}\) satellite in orbit \(150 \mathrm{~km}\) above Earth's...
-
Four objects are moved in various ways in the electrostatic field of Figure P25.19. Rank the following motions in order of the amount of electrostatic work done on the object, smallest amount first:...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the ERR for this project. b. Is this project economically attractive? EOY 0 2 3 4 5 6 NCF -$100 $15 $15 $15...
-
On a graph of the market for low-skilled labor, show the effect of the increase in the minimum wage on the quantity of labor employed. The rise in the federal minimum wage will boost the wages of the...
-
On a graph of the market for low-skilled labor, show the effect of the $26 a week increase in the minimum wage on employment of low-skilled labor. The government has increased the minimum wage by $26...
-
Bar Codes versus RFID You work as an efficiency analyst at one of the largest retail companies in the world, with multiple stores in every state, as well as in many other countries. For the past...
-
1-Stern observed all of the following results EXCEPT _______ in his experiment. A-one of the recombinant phenotypes was associated with an X chromosome of normal length B-the number of car, B+ male...
-
Reconsider the polymer viscosity data in Exercise 6.63. Use the median of the span-two moving ranges to estimate and set up the individuals control chart. Compare this chart to the one originally...
-
A normally distributed quality characteristic has specification limits at LSL = 10 and USL = 20. A random sample of size 50 results in x =16 and s =1.2 . (a) Calculate a point estimate of Cpk. (b)...
-
A belt that is used in a drive mechanism in a copier machine is required to have a minimum tensile strength of LSL = 150 lb. It is known from long experience that = 5 lb for this particular belt....
-
Alan, Bob and Charles are in partnership sharing profits and losses in the ratio 3:2:1 respectively. The statement of financial position for the partnership as at 30 June 2016 is as follows: Charles...
-
The statement of financial position of A. Barnes and C. Darwin at 31 March 2015 is as follows: The partners share profits and losses: Barnes three-fifths and Darwin two-fifths. At the date of the...
-
At 31 December 2017, the statement of financial position of A, B and C, who are equal partners, was as follows: A retired at that date. In order to determine the amount due to him the following...

Study smarter with the SolutionInn App


