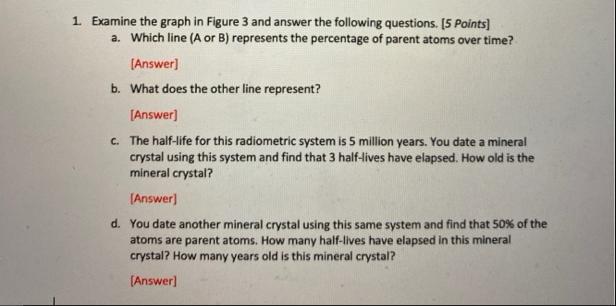
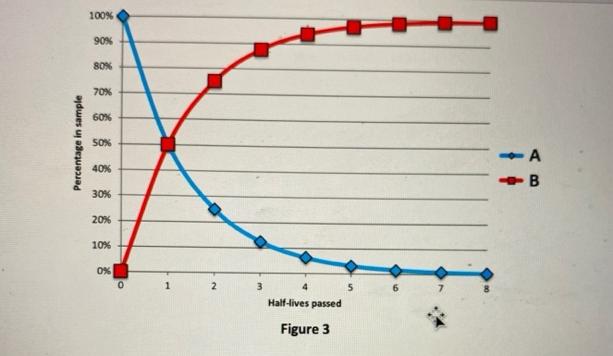
1. Examine the graph in Figure 3 and answer the following questions. [5 Points] a. Which...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1. Examine the graph in Figure 3 and answer the following questions. [5 Points] a. Which line (A or B) represents the percentage of parent atoms over time? [Answer] b. What does the other line represent? [Answer] c. The half-life for this radiometric system is 5 million years. You date a mineral crystal using this system and find that 3 half-lives have elapsed. How old is the mineral crystal? [Answer] d. You date another mineral crystal using this same system and find that 50% of the atoms are parent atoms. How many half-lives have elapsed in this mineral crystal? How many years old is this mineral crystal? [Answer] Percentage in sample 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% 0 1 2 3 Half-lives passed Figure 3 A -B 1. Examine the graph in Figure 3 and answer the following questions. [5 Points] a. Which line (A or B) represents the percentage of parent atoms over time? [Answer] b. What does the other line represent? [Answer] c. The half-life for this radiometric system is 5 million years. You date a mineral crystal using this system and find that 3 half-lives have elapsed. How old is the mineral crystal? [Answer] d. You date another mineral crystal using this same system and find that 50% of the atoms are parent atoms. How many half-lives have elapsed in this mineral crystal? How many years old is this mineral crystal? [Answer] Percentage in sample 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% 0 1 2 3 Half-lives passed Figure 3 A -B
Expert Answer:
Answer rating: 100% (QA)
Ans Line A represents the time of parent atoms over generally it represents the decay rate o... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these sociology questions
-
Examine the graph in Figure 21-1 and answer the following questions: (a) The area bounded by the lines AB, BC, CD, and AD is called the feasible area. Why? (b) The triangles BCF and CDE are not part...
-
Find all the articulation points in the graph in Figure 9.85. Show the depth-first spanning tree and the values of Num and Low for each vertex. K
-
The graph in Figure describes the acceleration as a function of time for a stone rolling down a hill starting from rest. (a) Find the change in the stone's velocity between t = 2.5 s and t = 7.5 s....
-
Q5] A firm has reported a profit of Rs.1,47,000 for the year ended 31-3-2014 after taking into consideration the following items. (i) The cost of an asset Rs.23,000 has been taken as an expense (ii)...
-
The Balanced, Inc., has three different product lines. Its least risky product line has a beta of 1.7, while its middle-risk product line has a beta of 1.8, and its most risky product line has a beta...
-
Los Angeles Lumber Company is considering a project with a cost of $1,000 initially, and inflows of $300 at the end of years 1-5. LALC's cost of capital is 12 percent. What is the project's IRR and...
-
Opposite are the comparative statements of financial position of Foxbell Ltd as at 30 June 2024 and 2025, and the statement of financial performance and statement of changes in equity for the year...
-
1. Describe the market segment toward which the ReadyMade magazine is primarily targeted. 2. What key attributes of ReadyMade magazine appeal to this market segment? 3. How is the target market of...
-
If f(x)=(8x3+9) and g(x) = -(9x + 19), then d dx [(2)-9(2)] -0
-
A distillation column can be purchased and installed for P 2,000,000. It will incur P35,000 in operations and maintenance expenses and yet can start generating revenues of P125,000 each month as soon...
-
Things were getting ugly in the cosmetic departments at Bon- Ton Department stores and something had to be done, fast. Beauty Advisors in their 277 stores were leaving the company at much higher...
-
York's outstanding stock consists of 80,000 shares of cumulative 7.5% preferred stock with a $5 par value and also 200,000 shares of common stock with a $1 par value. During its first four years of...
-
What assumption is NFPA 101 built upon with regards to the number of fire sources? Why might this be inadequate in the case of incendiary or set fires?
-
Write a program using Scanner and its nextLine method. The following is an example of how to use nextLine Scanner kybd = new Scanner(System.in); System.out.println("Enter a line of text"); String...
-
Bal., 1,600 units, 35% completed 17,440 To Finished Goods, 29,600 units ? Direct materials, 29,000 units @ 275,500 $9.50 Direct labor Factory overhead 84,600 39,258 Bal. ? units, 45% completed...
-
Demonstrate your understanding of memory system by designing a basic ARM memory system with the following information An ARM memory system has 4 physical 4Kx8 EPROM memory located at 0000h. There are...
-
You are considering the purchase of a commercial building. The current monthly rent income is $6,400. You anticipate being able to sell the building after 12 years for $975,000. If you require a 15%...
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
Scott Butterfield is self-employed as a CPA. He uses the cash method of accounting, and his Social Security number is 644-47-7833. His principal business code is 541211. Scott's CPA practice is...
-
For each of the following cases, indicate the filing status for the taxpayer(s) for 2012 using the following legend: A - Single B - Married, filing a joint return C - Married, filing separate returns...
-
William sold Section 1245 property for $25,000 in 2012. The property cost $35,000 when it was purchased 5 years ago. The depreciation claimed on the property was $16,000. a. Calculate the adjusted...
-
A mixture of benzene and monochlorobenzene is to be separated into almost pure products by distillation. Determine an appropriate operating pressure at the top of the tower.
-
The feed to a distillation tower consists of \(14.3 \mathrm{kmol} / \mathrm{hr}\) of methanol, \(105.3 \mathrm{kmol} / \mathrm{hr}\) of toluene, \(136.2 \mathrm{kmol} / \mathrm{hr}\) of ethylbenzene,...
-
In a reboiled absorber, operating as a deethanizer at 400 psia to separate a light hydrocarbon feed, conditions at the bottom tray are: Liquid Phase Molar flow = 1, \(366 \mathrm{lbmol} /...

Study smarter with the SolutionInn App


