Give the reagents and conditions required to convert 1-butyne into each of the following compounds (some...
Fantastic news! We've Found the answer you've been seeking!
Question:
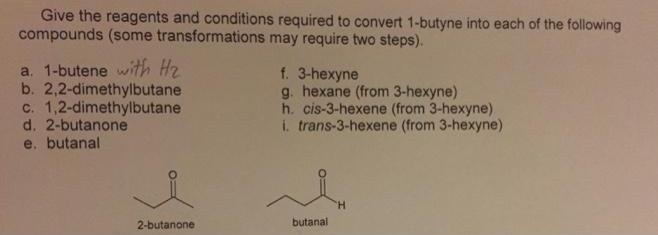
Transcribed Image Text:
Give the reagents and conditions required to convert 1-butyne into each of the following compounds (some transformations may require two steps). a. 1-butene with H2 b. 2,2-dimethylbutane c. 1,2-dimethylbutane d. 2-butanone e. butanal f. 3-hexyne g. hexane (from 3-hexyne) h. cis-3-hexene (from 3-hexyne). i. trans-3-hexene (from 3-hexyne) H. 2-butanone butanal Give the reagents and conditions required to convert 1-butyne into each of the following compounds (some transformations may require two steps). a. 1-butene with H2 b. 2,2-dimethylbutane c. 1,2-dimethylbutane d. 2-butanone e. butanal f. 3-hexyne g. hexane (from 3-hexyne) h. cis-3-hexene (from 3-hexyne). i. trans-3-hexene (from 3-hexyne) H. 2-butanone butanal
Expert Answer:
Answer rating: 100% (QA)
Solution H2 Lindlar Cat HC ECH but1ene 1 MeMgBr 2 H30 CH 1 PBr3 2 MgDry ether 3 CH3I HgS04 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Each of the following compounds has been prepared from o-anisidine (o-methoxyaniline). Outline a series of steps leading to each one. (a) o-Bromoanisole (d) 3-Fluoro-4-methoxybenzonitrile (b)...
-
Each of the following compounds is characterized by a 1H NMR spectrum that consists of only a single peak having the chemical shift indicated. Identify each compound. (a) C8H18; 0.9 ppm (f) C2H3Cl3;...
-
Each of the following compounds has three possible names listed for it. For each compound, what is the correct name and why arent the other names used? a. N2O: nitrogen oxide, nitrogen (I) oxide,...
-
Consider a set of documents. Assume that all documents have been normalized to have unit length of 1. What is the "shape" of a cluster that consists of all documents whose cosine similarity to a...
-
Show the effect of each of the following transactions on the basic accounting equation, by preparing a table like the one in Exhibit 2-3. The companys fiscal year end is December 31. a. On January 1,...
-
Each of the following aldehydes or ketones is known by a common name. Its substitutive IUPAC name is provided in parentheses. Write a structural formula for each one. (a) Chloral...
-
In 16 experiments studying the electrical behavior of single cells, 12 use micro-electrodes made of metal and the other 4 use micro-electrodes made from glass tubing. If 2 of the experiments are to...
-
John Smith has developed the following forecasting model: Y = 36 + 4.3X1 Where Y = Demand for K10 air conditioners X1 = the outside temperature (F) (a) Forecast the demand for K10 when the...
-
You are invited to a weekend carnival and behind your food truck is a large tree. The tree is down an embankment 100m away. You take measurement and want to know the height of the tree. Use the...
-
The Stratton Township Park is located on a piece of property that contains two golf courses, a swimming pool, and 800 acres of woods and open spaces. Three years ago, the Stratton Park Department...
-
Southern Company's accountant failed to accrue as of 12/31/13 some employee fringe benefit program expenses that were incurred in 2013 and that will be paid in 2014. The result of this omission is...
-
Describe the evidence that was used by Watson and Crick to define the double helix structure of DNA. and Explain
-
What role does organizational justice play in shaping employee attitudes towards change management initiatives? Explain
-
What are the 4 sources of constitutional law and their significance? (Canada)
-
Explain five principles of why business relationships are important?
-
Please define and discuss the term "ripeness" as it relates to constitutional law.
-
13. A metre-stick with a fulcrum and 2 loads as shown. F FL a. 1st class b. 2nd class c. 3rd class d. not a lever
-
Reconsider Prob. 1474. In order to drain the tank faster, a pump is installed near the tank exit as in Fig. P1475. Determine how much pump power input is necessary to establish an average water...
-
Tell whether the Ha and Hb hydrogens in each of the following compounds are enantiotopic, diastereotopic, or neither. a. b. c. d. CH3CH2CCH3 HV HI Br Hb H3C -
-
Draw the structures and name the four constitutional isomers with molecular formula C4H9 Br.
-
What alkyne would you start with and what reagents would you use if you wanted to synthesize: a. Pentane? b. Cis-2-butene? c. Trans-2-pentene? d. 1-hexene?
-
For the pediatrician presented in Example 1, find the probability that a randomly selected three-year-old girl is between 35 and 40 inches tall, inclusive. That is, find P(35 X 40). By-Hand...
-
The heights of a pediatricians three-year-old females are approximately normally distributed, with mean 38.72 inches and standard deviation 3.17 inches. Find the height of a three-year-old female at...
-
The scores earned on the mathematics portion of the SAT, a college entrance exam, are approximately normally distributed with mean 516 and standard deviation 116. What scores separate the middle 90%...

Study smarter with the SolutionInn App


