Question: 1. Glucose will react with oxygen in a laboratory setting using a combustion chamber. The reaction is exergonic, with the release of a large amount
1. Glucose will react with oxygen in a laboratory setting using a combustion chamber. The reaction is exergonic, with the release of a large amount of energy in the form of heat and the production of carbon dioxide and water according to the following chemical equation:
C 6 H 12 O 6 + 6O 2 ? 6CO 2 + 6H 2 O ? ? ? G = ?686 kcal/mol
Glucose can also be incubated with cells that completely oxidize glucose, according to the same net chemical equation and with the same quantity of energy release. However, glucose oxidation in cells occurs during a series of steps, rather than in one step like the combustion reaction taking place in a lab. The diagram below shows these steps in terms of the free energy change at each step, as well as the input and output of ATP, NADH, and FADH 2 at different stages of the process.
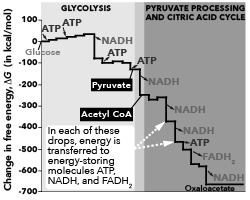
Free energy change and input/output during glucose oxidation
a. A coupled reaction is a pair of reactions occurring together in which the energy released by one reaction is used in bond formation in the second reaction. For example, glycolysis and pyruvate processing form a coupled reaction. Explain how coupling reactions allows a cell to capture the free energy of glucose oxidation rather than allow it to escape to the environment in the form of heat. Describe how this process affects other reactions in the cell. (3 points)
b. Pose a scientific question that a scientist might ask about how these processes are conserved across phyla. (3 points)
Change in free energy, AG (in kcal/mol) 100- Glucose -100- -200- -300- GLYCOLYSIS ATP ATP NADH ATP ATP Pyruvate NADH Acetyl CoA 400 In each of these 500 -600- -700 PYRUVATE PROCESSING AND CITRIC ACID CYCLE drops, energy is transferred to energy-storing molecules ATP, NADH, and FADH NADH NADH -ATP FADH NADH Oxaloacetate
Step by Step Solution
3.41 Rating (145 Votes )
There are 3 Steps involved in it
a Coupling Reactions in Cells Explanation of Coupling Reactions 1 Energy Transfer Coupling reactions ... View full answer

Get step-by-step solutions from verified subject matter experts


