1. Predict the product(s) and provide the mechanism for each reaction below. (CH,CH),NH H* Catalyst b....
Fantastic news! We've Found the answer you've been seeking!
Question:
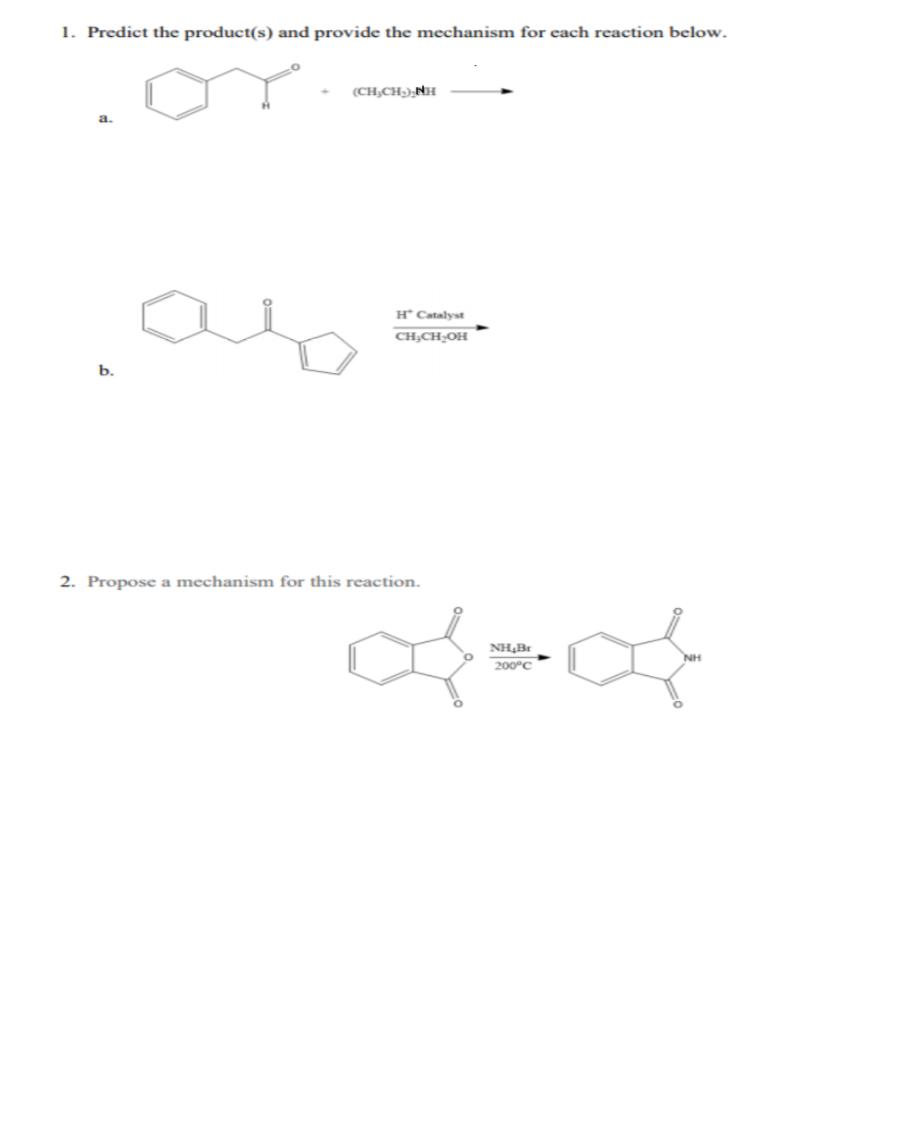
Transcribed Image Text:
1. Predict the product(s) and provide the mechanism for each reaction below. (CH,CH),NH H* Catalyst b. 2. Propose a mechanism for this reaction. NH,Br NH 200°C 1. Predict the product(s) and provide the mechanism for each reaction below. (CH,CH),NH H* Catalyst b. 2. Propose a mechanism for this reaction. NH,Br NH 200°C
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
1. Observe the timing diagram below for a certain type of circuit, with inputs A, B, C, D, and output G. a. Express G as a minimal SOP in terms of A, B, C and D. [4] b. Briefly describe the function...
-
Propose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of a peroxide. ROOR- H2C CHCH high pressure C-C propylene I7 polypropylene
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Find all values of 0, if 0 is in the interval [0, 360) and has the given function value. cot 0= -1 0= (Type your answer in degrees. Use a comma to separate answers as needed.)
-
A new manager has been hired to run the campus student pub. The pub sells a limited selection of brands of beer. The past manager indicated that the brand preferences of the pub's customers were as...
-
Not all hotel owners enter into franchise agreements. Why do you think a hotel owner might choose not to affiliate with a franchise brand?
-
For fluids with \(\operatorname{Pr} <1\) the velocity profile is assumed to be a cubic for \(y
-
Your client is 40 years old; and she wants to begin saving for retirement, with the first payment to come one year from now. She can save $5,000 per year; and you advise her to invest it in the stock...
-
An 80.0 kg sprinter starts a race with an acceleration of 1.96 m/s2. If the sprinter accelerates at that rate for 34 m, and then maintains that velocity for the remainder of the 100 m dash, what will...
-
A work sheet for Juanita's Consulting is shown on the following page. There were no additional investments made by the owner during the month. REQUIRED 1. Prepare an income statement. 2. Prepare a...
-
Discuss some strategies and types of information and coordination in regards to communication that effective emergency planners would want to have in place prior to an event.
-
Construct Histograms for each of the following values. Team Revenue ($mil) Value ($mil) Manchester United 765 3689 Real Madrid 688 3580 Arsenal 520 1932 Barcelona 688 3636 Bayern Munich 657 2713...
-
Use GM file data that is listed below. GM data is a 5-year stock price and trade volume for General Motors Company (GM) from May 2016 through April 2021. This data includes average daily open, high,...
-
A 2x8 joist (b=1.5"; h-7.25") is loaded with a uniform load (w) of 60 #/ft on a simple 12' span. Determine the maximum bending stress (fb) and the maximum shear stress (fv) developed in the joist....
-
Natalie is thinking of buying a van that will only be used for her business, Baking Time. The cost of the van is $41,000. Natalie would spend an additional $3,000 to have the van painted with the...
-
Find: a. Cumalative Frequencies for each score. b. Percentile Ranks for each score. c. The 5th, 15th, 30th, 85th, and 95th Percentiles. d. The First, Second and Third Percentiles, Q Q2 Q3...
-
Given the following information, calculate the cash flow statement for the current and prior year using the indirect method. Exhibit 1: Income Statement (US$ Thousands) Current year Prior year Two...
-
Chapter 9 Stock Valuation at Ragan Engines Input area: Shares owned by each sibling Ragan EPS Dividend to each sibling Ragan ROE Ragan required return Blue Ribband Motors Corp. Bon Voyage Marine,...
-
Give equations for the formation and hydrogenolysis of glutamine benzyl ester.
-
Addition of 1-bromobut-2-ene to magnesium metal in dry ether results in formation of a Grignard reagent. Addition of water to this Grignard reagent gives a mixture of but-1-ene and but-2-ene (cis and...
-
Using what you know about the conformational energetics of substituted cyclohexanes, predict which of the two decalin isomers is more stable. Explain your reasoning.
-
Personalized medicine may be used a. to characterize types of tumors. b. to predict the outcome of certain types of cancers. c. to determine the proper dosage of drugs. d. in all of the above.
-
Tumor-suppressor genes promote cancer when a. they are overexpressed. b. they are expressed in the wrong cell type. c. their function is inactivated. d. they are expressed at the wrong stage of...
-
Which of the following is a type of genetic change that could produce an oncogene? a. Missense mutation b. Gene amplification c. Chromosomal translocation d. All of the above can produce an oncogene.

Study smarter with the SolutionInn App



