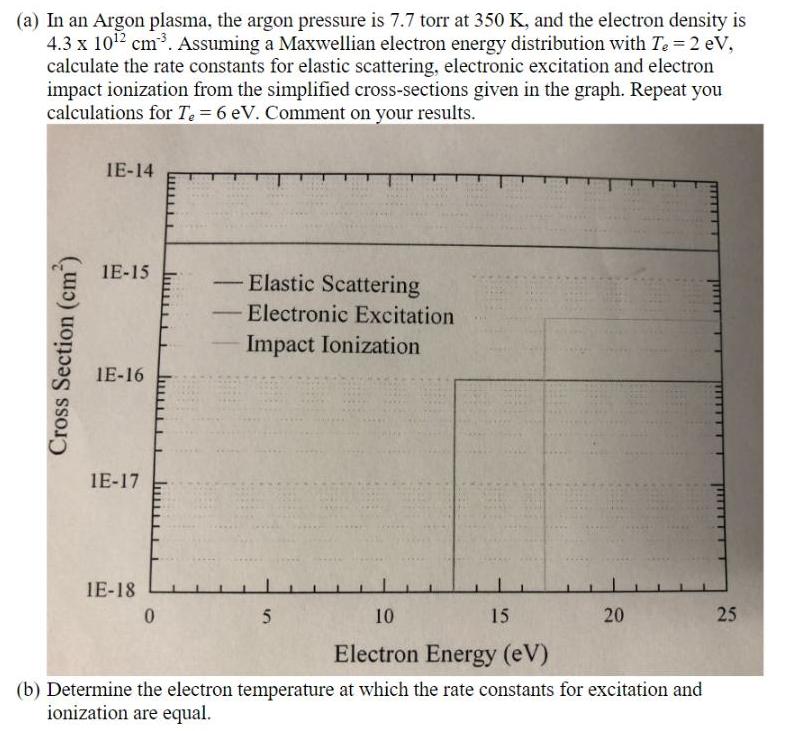
(a) In an Argon plasma, the argon pressure is 7.7 torr at 350 K, and the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(a) In an Argon plasma, the argon pressure is 7.7 torr at 350 K, and the electron density is 4.3 x 1012 cm3. Assuming a Maxwellian electron energy distribution with Te 2 eV, calculate the rate constants for elastic scattering, electronic excitation and electron impact ionization from the simplified cross-sections given in the graph. Repeat you calculations for Te = 6 eV. Comment on your results. IE-14 1E-15 Elastic Scattering - Electronic Excitation - Impact Ionization IE-16 1E-17 IE-18 10 15 20 25 Electron Energy (eV) (b) Determine the electron temperature at which the rate constants for excitation and ionization are equal. Cross Section (cm) (a) In an Argon plasma, the argon pressure is 7.7 torr at 350 K, and the electron density is 4.3 x 1012 cm3. Assuming a Maxwellian electron energy distribution with Te 2 eV, calculate the rate constants for elastic scattering, electronic excitation and electron impact ionization from the simplified cross-sections given in the graph. Repeat you calculations for Te = 6 eV. Comment on your results. IE-14 1E-15 Elastic Scattering - Electronic Excitation - Impact Ionization IE-16 1E-17 IE-18 10 15 20 25 Electron Energy (eV) (b) Determine the electron temperature at which the rate constants for excitation and ionization are equal. Cross Section (cm)
Expert Answer:
Answer rating: 100% (QA)
Rate Cooficients and Co s Sactions a For vamy genekal olision fuocess the ecte coefficient camy ... View the full answer

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these chemistry questions
-
An electron with kinetic energy T 4 eV is confined within a region whose linear dimension is l = 1m. Using the uncertainty principle, evaluate the relative uncertainty of its velocity.
-
At Stevens Manufacturing, workers are organized in teams that combine their efforts to produce complex units of a final product. Stevens is using a facility layout known as a(n): consolidated process...
-
Calculate the mode, mean, and median of the following data: 18 10 15 13 17 15 12 15 18 16 11
-
A fast-food restaurant operates both a drive through facility and a walk-in facility. On a randomly selected day, let X and Y, respectively, be the proportions of the time that the drive-through and...
-
Is there a preferred sequence of steps? Why?
-
Explain which compound has the higher melting point or boiling point: a) Melting point b) Boiling point or C COCH c) Boiling point or
-
Why is duration important?
-
Project managers must not only calculate variances but also determine the root cause of the variance. Some variances may be allowable while others must be explained together with a corrective plan...
-
16. A signal containing multiple frequencies is shown in figure A. It is passed through 2 different filters and then outputs are shown in figure B & C. M (A) S (B) sha (C) (a) B corresponds to low...
-
The university computer lab's director keeps track of the lab usage, as measured by the number of students using the lab. This particular function is very important for budgeting purposes. The...
-
W. Deng and K. W. Hipps (J. Phys. Chem. B 2003, 107, 10736-10740) reported an STM study of the electronic properties of Ni(ll)tetraphenyl porphyrin (NiTPP), a red-purple, neutral diamagnetic complex...
-
a) b) 308 Suppose SSH Inc. is currently $12 per share and will go either up to $16 or down to $10. Suppose the risk-free interest rate is 3%. We are interested in finding the price of a put option....
-
Use Newton's method to approximate a root of the equation 3x7 + 4x4 + 2 = 0 as follows. Let x1 = 2 be the initial approximation. The second approximation 2 is and the third approximation 23 is Carry...
-
You have done the Innovation Report and recommended supporting an inventive workplace, and You emailed the report to your manager and the company's owners for review. A meeting has been planned with...
-
Service providers that produce goods can, like manufacturers, adopt either a make-to-order or a make-to-stock approach to manufacturing them. BK, which encourages patrons to customize burgers and...
-
A company has a 10 year annual return of 7,65% and standard deviation of 13,24%. I calculated AM( 0,1058) and GM( 0,0986)of the market index over 10 years. The market risk i found was 13,12% and co...
-
You are a preschool director of a prestigious school, with a long waiting list. You are fortunate because a wealthy benefactor donates a lot of money to your school each year; allowing you to always...
-
In the simple quantity theory of money, what will lead to an increase in aggregate demand? In monetarism, what will lead to an increase in aggregate demand?
-
Consider the decay A0 p + - with the A0 at rest. (a) Calculate the disintegration energy. What is the kinetic energy of? (b) The proton and (c) The pi on?
-
A thin rod of length 0.75 m and mass 0.42 kg is suspended freely from one end. It is pulled to one side and then allowed to swing like a pendulum, passing through its lowest position with angular...
-
The maximum force you can exert on an object with one of your back teeth is about 750 N. Suppose that as you gradually bite on a clump of licorice, the licorice resists compression by one of your...
-
Diehl Cleaners has the following statement of financial position items. Instructions Classify each item as an asset, liability, or equity. Analyze the effect of transactions. Accounts payable Cash...
-
Erin Danielle, the bookkeeper for Liverpool Ltd., has been trying to determine the correct statement of financial position for the company. The companys statement of financial position is shown...
-
Which of the following is true? a. Financial frauds have not occurred in U.S. companies because GAAP has detailed accounting and disclosure requirements. b. Transaction analysis is basically the same...

Study smarter with the SolutionInn App


