1. Which of the following statements is incorrect about benzene? A) All of the carbon atoms...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
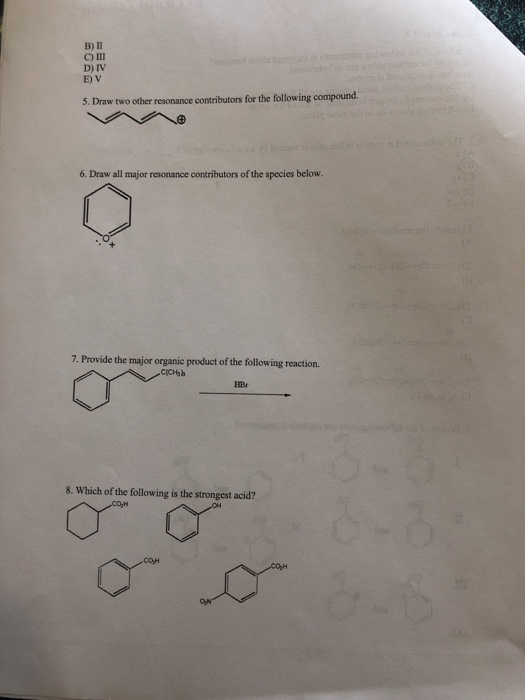
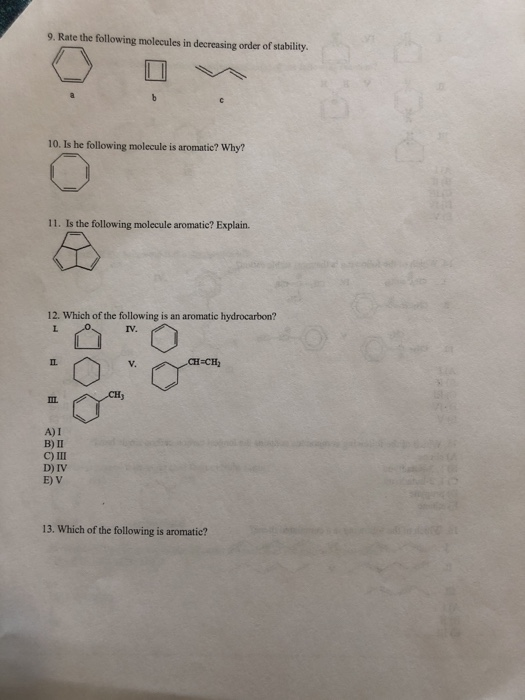
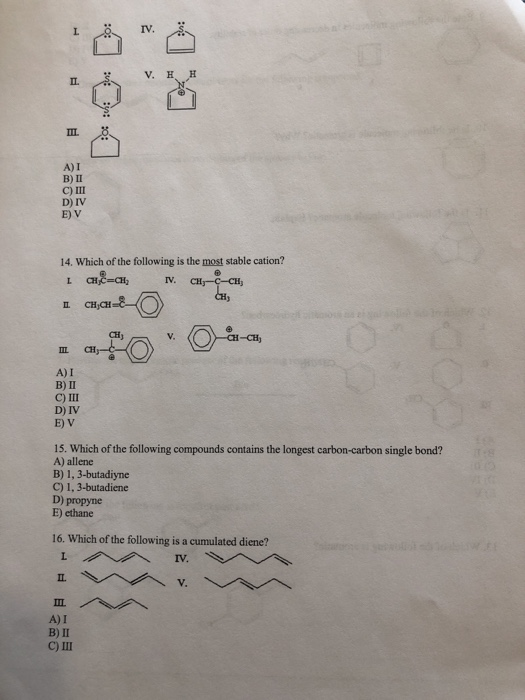
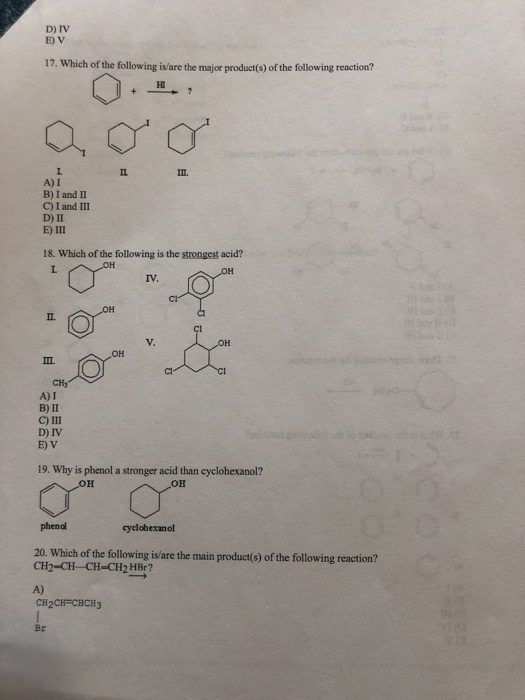
1. Which of the following statements is incorrect about benzene? A) All of the carbon atoms are sp hybridized. B) It has delocalized electrons. C) The carbon-carbon bond lengths are all the same. D) The carbon-hydrogen bond lengths are all the same. E) All twelve atoms lie in the same plane. 2. The delocalized system in benzene is formed by a cyclic overlap of 6 A) s (B) C) sp D) sp2 E) sp3 3.Identify the resonance hybrid. A) ✪ CH₂CH=CH-CH=CH-CH₂ ✪ CH₂CH=CH-CH-CH=CH₂ B) C) CH₂CH-CH=CH-CH=CH₂ D) Ⓒ 8+ 8+ CH₂CH=CH-CH==CH==CH₂ E) A, B, and C. II. 4. Which of the following pairs are resonance structures? CH3 CH3 8-6 and III. 8+ A) I CH₂ CH₂ 8-6 and CH3 CH₂ &-$ and IV. V. CH3 Ⓒ and and CH₂ CH₂ orbitals. W B) II C) III D) IV E) V 5. Draw two other resonance contributors for the following compound. 6. Draw all major resonance contributors of the species below. 7. Provide the major organic product of the following reaction. -C(CH₂) HBr 8. Which of the following is the strongest acid? CO₂H H 3-3 9. Rate the following molecules in decreasing order of stability. 10. Is he following molecule is aromatic? Why? 11. Is the following molecule aromatic? Explain. 12. Which of the following is an aromatic hydrocarbon? L IV. II. III. A) I B) II C) III D) IV E) V CH₂ V. CH=CH₂ 13. Which of the following is aromatic? III. A) I B) II C) III D) IV E) V II. III 14. Which of the following is the most stable cation? L CH₂=CH₂ IV. ICH₂CH= A) I B) II C) III D) IV E) V E CH₂- B) 1, 3-butadiyne C) 1,3-butadiene D) propyne E) ethane II. CH₂ IV. III A) I B) II C) III V. H. H 15. Which of the following compounds contains the longest carbon-carbon single bond? A) allene V. 16. Which of the following is a cumulated diene? L IV. CH₂-C-CH₂ CH₂ -CH-CH₂ V. 10 1:8 D) IV E) V 17. Which of the following is/are the major product(s) of the following reaction? HI L A) I B) I and II C) I and III D) II E) III II. E III. 18. Which of the following is the strongest acid? L OH OH A) I B) II C) III D) IV E) V A) CH₂ phenol IL OH CH2CH=CHCH3 Br + OH IV. ? V. III. 19. Why is phenol a stronger acid than cyclohexanol? OH OH CI cyclohexanol 20. Which of the following is/are the main product(s) of the following reaction? CH2=CH-CH=CH2 HBr? OH CI VOL B) CH₂CH₂CH=CH2 F 1 Br C) III. CH3CHCH=CH2 Br D) A and B E) A and C 21. What are the products from the following reaction? + Bry/CCL -? L A) I and II B) I and III II. C) I and IV D) II and III E) II and IV 22. Draw the product(s) for the reaction. HC A) I B) II -CH₂CH₂ 23. What is the product of the following reaction? beat C) III D) IV E) V III. IV. Be IV. ? 24. What is the product from the following reaction? COOK heat L II. III. A) I B) II C) III D) IV E) V II III. COOK A) I B) II C) III D) IV E) V COOK COOK COOK COOH COOK "COOH IV. V. V. 25. Which of the following dienes is the most reactive in a Diels-Alder reaction? I. IV. 7 COOK COOH COOK 1. Which of the following statements is incorrect about benzene? A) All of the carbon atoms are sp hybridized. B) It has delocalized electrons. C) The carbon-carbon bond lengths are all the same. D) The carbon-hydrogen bond lengths are all the same. E) All twelve atoms lie in the same plane. 2. The delocalized system in benzene is formed by a cyclic overlap of 6 A) s (B) C) sp D) sp2 E) sp3 3.Identify the resonance hybrid. A) ✪ CH₂CH=CH-CH=CH-CH₂ ✪ CH₂CH=CH-CH-CH=CH₂ B) C) CH₂CH-CH=CH-CH=CH₂ D) Ⓒ 8+ 8+ CH₂CH=CH-CH==CH==CH₂ E) A, B, and C. II. 4. Which of the following pairs are resonance structures? CH3 CH3 8-6 and III. 8+ A) I CH₂ CH₂ 8-6 and CH3 CH₂ &-$ and IV. V. CH3 Ⓒ and and CH₂ CH₂ orbitals. W B) II C) III D) IV E) V 5. Draw two other resonance contributors for the following compound. 6. Draw all major resonance contributors of the species below. 7. Provide the major organic product of the following reaction. -C(CH₂) HBr 8. Which of the following is the strongest acid? CO₂H H 3-3 9. Rate the following molecules in decreasing order of stability. 10. Is he following molecule is aromatic? Why? 11. Is the following molecule aromatic? Explain. 12. Which of the following is an aromatic hydrocarbon? L IV. II. III. A) I B) II C) III D) IV E) V CH₂ V. CH=CH₂ 13. Which of the following is aromatic? III. A) I B) II C) III D) IV E) V II. III 14. Which of the following is the most stable cation? L CH₂=CH₂ IV. ICH₂CH= A) I B) II C) III D) IV E) V E CH₂- B) 1, 3-butadiyne C) 1,3-butadiene D) propyne E) ethane II. CH₂ IV. III A) I B) II C) III V. H. H 15. Which of the following compounds contains the longest carbon-carbon single bond? A) allene V. 16. Which of the following is a cumulated diene? L IV. CH₂-C-CH₂ CH₂ -CH-CH₂ V. 10 1:8 D) IV E) V 17. Which of the following is/are the major product(s) of the following reaction? HI L A) I B) I and II C) I and III D) II E) III II. E III. 18. Which of the following is the strongest acid? L OH OH A) I B) II C) III D) IV E) V A) CH₂ phenol IL OH CH2CH=CHCH3 Br + OH IV. ? V. III. 19. Why is phenol a stronger acid than cyclohexanol? OH OH CI cyclohexanol 20. Which of the following is/are the main product(s) of the following reaction? CH2=CH-CH=CH2 HBr? OH CI VOL B) CH₂CH₂CH=CH2 F 1 Br C) III. CH3CHCH=CH2 Br D) A and B E) A and C 21. What are the products from the following reaction? + Bry/CCL -? L A) I and II B) I and III II. C) I and IV D) II and III E) II and IV 22. Draw the product(s) for the reaction. HC A) I B) II -CH₂CH₂ 23. What is the product of the following reaction? beat C) III D) IV E) V III. IV. Be IV. ? 24. What is the product from the following reaction? COOK heat L II. III. A) I B) II C) III D) IV E) V II III. COOK A) I B) II C) III D) IV E) V COOK COOK COOK COOH COOK "COOH IV. V. V. 25. Which of the following dienes is the most reactive in a Diels-Alder reaction? I. IV. 7 COOK COOH COOK 1. Which of the following statements is incorrect about benzene? A) All of the carbon atoms are sp hybridized. B) It has delocalized electrons. C) The carbon-carbon bond lengths are all the same. D) The carbon-hydrogen bond lengths are all the same. E) All twelve atoms lie in the same plane. 2. The delocalized system in benzene is formed by a cyclic overlap of 6 A) s (B) C) sp D) sp2 E) sp3 3.Identify the resonance hybrid. A) ✪ CH₂CH=CH-CH=CH-CH₂ ✪ CH₂CH=CH-CH-CH=CH₂ B) C) CH₂CH-CH=CH-CH=CH₂ D) Ⓒ 8+ 8+ CH₂CH=CH-CH==CH==CH₂ E) A, B, and C. II. 4. Which of the following pairs are resonance structures? CH3 CH3 8-6 and III. 8+ A) I CH₂ CH₂ 8-6 and CH3 CH₂ &-$ and IV. V. CH3 Ⓒ and and CH₂ CH₂ orbitals. W B) II C) III D) IV E) V 5. Draw two other resonance contributors for the following compound. 6. Draw all major resonance contributors of the species below. 7. Provide the major organic product of the following reaction. -C(CH₂) HBr 8. Which of the following is the strongest acid? CO₂H H 3-3 9. Rate the following molecules in decreasing order of stability. 10. Is he following molecule is aromatic? Why? 11. Is the following molecule aromatic? Explain. 12. Which of the following is an aromatic hydrocarbon? L IV. II. III. A) I B) II C) III D) IV E) V CH₂ V. CH=CH₂ 13. Which of the following is aromatic? III. A) I B) II C) III D) IV E) V II. III 14. Which of the following is the most stable cation? L CH₂=CH₂ IV. ICH₂CH= A) I B) II C) III D) IV E) V E CH₂- B) 1, 3-butadiyne C) 1,3-butadiene D) propyne E) ethane II. CH₂ IV. III A) I B) II C) III V. H. H 15. Which of the following compounds contains the longest carbon-carbon single bond? A) allene V. 16. Which of the following is a cumulated diene? L IV. CH₂-C-CH₂ CH₂ -CH-CH₂ V. 10 1:8 D) IV E) V 17. Which of the following is/are the major product(s) of the following reaction? HI L A) I B) I and II C) I and III D) II E) III II. E III. 18. Which of the following is the strongest acid? L OH OH A) I B) II C) III D) IV E) V A) CH₂ phenol IL OH CH2CH=CHCH3 Br + OH IV. ? V. III. 19. Why is phenol a stronger acid than cyclohexanol? OH OH CI cyclohexanol 20. Which of the following is/are the main product(s) of the following reaction? CH2=CH-CH=CH2 HBr? OH CI VOL B) CH₂CH₂CH=CH2 F 1 Br C) III. CH3CHCH=CH2 Br D) A and B E) A and C 21. What are the products from the following reaction? + Bry/CCL -? L A) I and II B) I and III II. C) I and IV D) II and III E) II and IV 22. Draw the product(s) for the reaction. HC A) I B) II -CH₂CH₂ 23. What is the product of the following reaction? beat C) III D) IV E) V III. IV. Be IV. ? 24. What is the product from the following reaction? COOK heat L II. III. A) I B) II C) III D) IV E) V II III. COOK A) I B) II C) III D) IV E) V COOK COOK COOK COOH COOK "COOH IV. V. V. 25. Which of the following dienes is the most reactive in a Diels-Alder reaction? I. IV. 7 COOK COOH COOK 1. Which of the following statements is incorrect about benzene? A) All of the carbon atoms are sp hybridized. B) It has delocalized electrons. C) The carbon-carbon bond lengths are all the same. D) The carbon-hydrogen bond lengths are all the same. E) All twelve atoms lie in the same plane. 2. The delocalized system in benzene is formed by a cyclic overlap of 6 A) s (B) C) sp D) sp2 E) sp3 3.Identify the resonance hybrid. A) ✪ CH₂CH=CH-CH=CH-CH₂ ✪ CH₂CH=CH-CH-CH=CH₂ B) C) CH₂CH-CH=CH-CH=CH₂ D) Ⓒ 8+ 8+ CH₂CH=CH-CH==CH==CH₂ E) A, B, and C. II. 4. Which of the following pairs are resonance structures? CH3 CH3 8-6 and III. 8+ A) I CH₂ CH₂ 8-6 and CH3 CH₂ &-$ and IV. V. CH3 Ⓒ and and CH₂ CH₂ orbitals. W B) II C) III D) IV E) V 5. Draw two other resonance contributors for the following compound. 6. Draw all major resonance contributors of the species below. 7. Provide the major organic product of the following reaction. -C(CH₂) HBr 8. Which of the following is the strongest acid? CO₂H H 3-3 9. Rate the following molecules in decreasing order of stability. 10. Is he following molecule is aromatic? Why? 11. Is the following molecule aromatic? Explain. 12. Which of the following is an aromatic hydrocarbon? L IV. II. III. A) I B) II C) III D) IV E) V CH₂ V. CH=CH₂ 13. Which of the following is aromatic? III. A) I B) II C) III D) IV E) V II. III 14. Which of the following is the most stable cation? L CH₂=CH₂ IV. ICH₂CH= A) I B) II C) III D) IV E) V E CH₂- B) 1, 3-butadiyne C) 1,3-butadiene D) propyne E) ethane II. CH₂ IV. III A) I B) II C) III V. H. H 15. Which of the following compounds contains the longest carbon-carbon single bond? A) allene V. 16. Which of the following is a cumulated diene? L IV. CH₂-C-CH₂ CH₂ -CH-CH₂ V. 10 1:8 D) IV E) V 17. Which of the following is/are the major product(s) of the following reaction? HI L A) I B) I and II C) I and III D) II E) III II. E III. 18. Which of the following is the strongest acid? L OH OH A) I B) II C) III D) IV E) V A) CH₂ phenol IL OH CH2CH=CHCH3 Br + OH IV. ? V. III. 19. Why is phenol a stronger acid than cyclohexanol? OH OH CI cyclohexanol 20. Which of the following is/are the main product(s) of the following reaction? CH2=CH-CH=CH2 HBr? OH CI VOL B) CH₂CH₂CH=CH2 F 1 Br C) III. CH3CHCH=CH2 Br D) A and B E) A and C 21. What are the products from the following reaction? + Bry/CCL -? L A) I and II B) I and III II. C) I and IV D) II and III E) II and IV 22. Draw the product(s) for the reaction. HC A) I B) II -CH₂CH₂ 23. What is the product of the following reaction? beat C) III D) IV E) V III. IV. Be IV. ? 24. What is the product from the following reaction? COOK heat L II. III. A) I B) II C) III D) IV E) V II III. COOK A) I B) II C) III D) IV E) V COOK COOK COOK COOH COOK "COOH IV. V. V. 25. Which of the following dienes is the most reactive in a Diels-Alder reaction? I. IV. 7 COOK COOH COOK
Expert Answer:

Related Book For 

Auditing and Assurance Services
ISBN: 978-0077862343
6th edition
Authors: Timothy Louwers, Robert Ramsay, David Sinason, Jerry Straws
Posted Date:
Students also viewed these chemistry questions
-
Which of the following statements is INCORRECT about market portfolio? A. Market portfolio is a hypothetical bundle of investments. B. It generally includes every type of assets that are available in...
-
Which of the following statements is incorrect regarding manufacturing overhead? a. Manufacturing overhead is an indirect costs to units or products. b. Manufacturing overhead includes both fixed and...
-
Which of the following statements is incorrect concerning an adjusted trial balance? a. An adjusted trial balance lists account balances and their locations in the ledger. b. An adjusted trial...
-
Giant Corporation (a U.S. corporation) forms Small Corporation in a foreign country. Giant owns 70% of Small Corporation's stock and the remaining stock is owned by citizens of the country in which...
-
Holman Electronics manufactures audio equipment, selling it through various distributors. Holmans days sales outstanding (Accounts receivable / Average daily credit sales) figures increased steadily...
-
Use an aggregate supply-and-demand diagram to study what would happen to an economy in which the aggregate supply curve never moved while the aggregate demand curve shifted outward year after year.
-
If your instructor assigns a marketing plan for your class, we hope you will be excitedfor two reasons. First, you will get insights into trying to actually do marketing that often go beyond what you...
-
The following data were taken from the balance sheet accounts of Wickham Corporation on December 31, 2010. Prepare the required journal entries for the following unrelated items. (a) A 5% stock...
-
An Olympic lifter (m =103kg) is holding a lift with a mass of 350 kg. The bar exerts a purely vertical force that is equally distributed between both hands. Each arm has a mass of 9 kg, are 0.8m long...
-
On March 1, Marigold Inc sells 1,400 common shares to its employees at $26 per share and lends the money to the employees to buy the new shares in exchange for a note receivable The employees pay 45...
-
How long (in years) will it take to quadruple a money if it earns 0.07 compounded semiannually? The given interest is already in decimal form. Do not round in between solution. Final answer round to...
-
2. Write a python function called nested_sum that takes a list of lists and adds up the elements from all of the nested lists. (5 marks) For example: >>> t = [[1, 2], [3], ['a','b'], [0.23,'e'], [4,...
-
What trade theory, policy or concepts were involved in the live export ban
-
Assume that the risk adjusted cost of capital is 16% for project A, 18% for project B, and 20% for project C. Project A: Estimated cash flow is $25,800 per year for 10 years. Cost $100,000 Project...
-
An initial investment of $41,800 fifty years ago is worth $1,533,913 today. Determine the annual return on this investment.
-
Consider the unbounded queue implementation shown in Fig. 10.21. This queue is blocking, meaning that the deq () method does not return until it has found an item to dequeu The queue has two fields:...
-
Smithson Pet Supply, Inc, had no noncash investing and finanang transactions during 2018. During the year, Smithson Pel Supply, Inc, sold no land or equipment issued no notes payable, retired no...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
This question contains three items that are management assertions about property and equipment. Following them are several substantive procedures for obtaining evidence about managements assertions....
-
Accounts Payable Confirmations. Partners Clark and Kent, both CPAs, are preparing their audit plan for the audit of accounts payable on Marlboro Corporations annual audit. Saturday afternoon they...
-
What type of opinion(s) would the audit team issue on the effectiveness of internal control over financial reporting if a material weakness in internal control exists? How would the standard report...
-
Generalize the model considered in Example 4.11 to a marginal model for the longitudinal DOS data and compare the findings with that in Example4.11 Example 4.11 For the models in Example 4.8 DOS,...
-
For the DTS study, use subjects with all five assessments in HamD scores in the CAU group for this question. The intraclass correlation coefficient among the repeated measures in Ham-D scores can be...
-
In this question we develop a regression model to assess the treatment effect for stigma in the DTS study, controlling for demographics and baseline measurements. We will use the cumulative logit...

Study smarter with the SolutionInn App


