Question: A treatment plant uses a two-stage suspended growth bioreactor to accomplish denitrification and removal of organic substrates. The first stage is anoxic, and denitrification
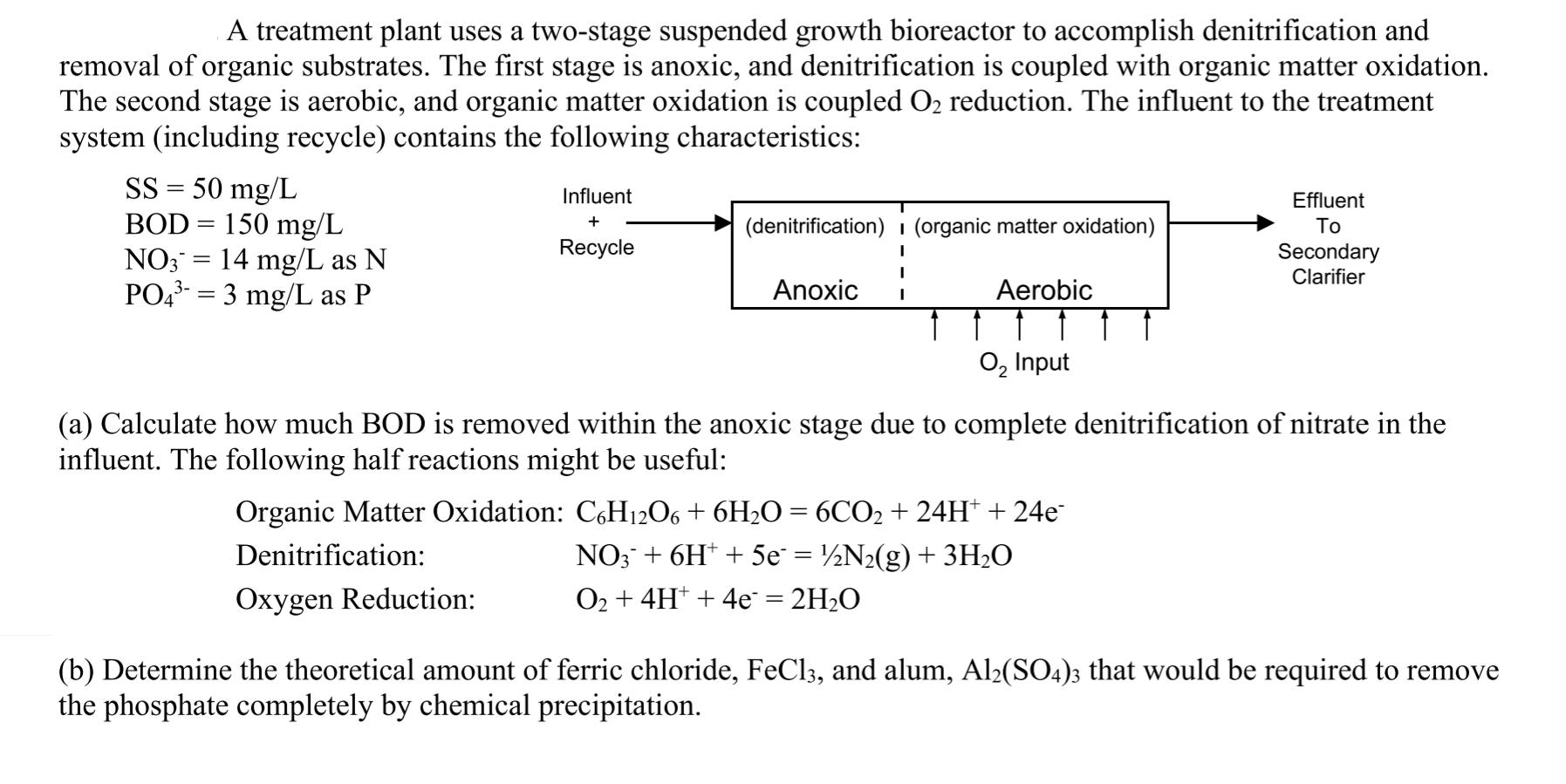
A treatment plant uses a two-stage suspended growth bioreactor to accomplish denitrification and removal of organic substrates. The first stage is anoxic, and denitrification is coupled with organic matter oxidation. The second stage is aerobic, and organic matter oxidation is coupled O reduction. The influent to the treatment system (including recycle) contains the following characteristics: SS = 50 mg/L BOD= 150 mg/L NO3 = 14 mg/L as N PO4-= 3 mg/L as P Influent + Recycle (denitrification) (organic matter oxidation) Anoxic I Aerobic Effluent To Secondary Clarifier O Input (a) Calculate how much BOD is removed within the anoxic stage due to complete denitrification of nitrate in the influent. The following half reactions might be useful: Organic Matter Oxidation: C6H12O6 + 6HO = 6CO + 24H+ + 24e Denitrification: NO3 + 6H+ + 5e = N(g) + 3HO Oxygen Reduction: O + 4H+ + 4e = 2HO (b) Determine the theoretical amount of ferric chloride, FeCl3, and alum, Al(SO4)3 that would be required to remove the phosphate completely by chemical precipitation.
Step by Step Solution
3.50 Rating (150 Votes )
There are 3 Steps involved in it
Following are the calculation for units to be manuf... View full answer

Get step-by-step solutions from verified subject matter experts


