1.00 kg of liquid water at 20.0 C is converted to superheated steam at 1200 C...
Fantastic news! We've Found the answer you've been seeking!
Question:
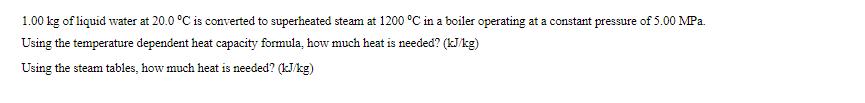
Transcribed Image Text:
1.00 kg of liquid water at 20.0 °C is converted to superheated steam at 1200 °C in a boiler operating at a constant pressure of 5.00 MPa. Using the temperature dependent heat capacity formula, how much heat is needed? (kJ/kg) Using the steam tables, how much heat is needed? (kJ/kg) 1.00 kg of liquid water at 20.0 °C is converted to superheated steam at 1200 °C in a boiler operating at a constant pressure of 5.00 MPa. Using the temperature dependent heat capacity formula, how much heat is needed? (kJ/kg) Using the steam tables, how much heat is needed? (kJ/kg)
Expert Answer:
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these mechanical engineering questions
-
A piston/cylinder assembly contains 1 kg of liquid water at 20oC and 300 kPa. There is a linear spring mounted on the piston such that when the water is heated the pressure reaches 1 MPa with a...
-
A piston/cylinder contains 1 kg of liquid water at 20C and 300 kPa. There is a linear spring mounted on the piston such that when the water is heated the pressure reaches 3 MPa with a volume...
-
A piston/cylinder assembly contains 1 kg of liquid water at 20oC and 300 kPa. There is a linear spring mounted on the piston such that when the water is heated the pressure reaches 1 MPa with a...
-
Let Z[] be the following set of rational numbers { a Z, k N} (and recall that N = {0, 1, 2,...} in this class). Let the addition and multiplication for Z[] be the usual addition and multiplication...
-
Determine the radius of curvature of the path described by the particle of Prob. 11.98 when t = 0, A = 3, and B = 1.
-
Why is it important to understand the nature of the clients business when conducting an audit of financial statement accounts?
-
When to file a motion to strike?
-
A random sample of 20 independent female college-aged dancers was obtained, and their heights (in inches) were measured. Assume the population distribution is Normal. a. What is the sample mean? Is...
-
What is the key problem in the case study Under Armour: Creating and Growing a New Consumer Brand. What are two solutions to the problems. What are advantages and disadvantages.
-
Elizabeth is moving from a one bedroom apartment in one city to a similar apartment in another city. She has been quoted a flat fee for the truck rental and has two estimates for wages of the movers...
-
Why would different industries need different types of business plans? - Vishal and Dilpreet Different costs of business in different industries and Different budgets Different target markets ...
-
When sampling is used to test the hypothesis that the evidence supports the account balance, what are the two parts of the sampling objective?
-
Why is it helpful to follow a project management methodology? What do you think about agile project management?
-
Matthews Corporation has changed from a system of recording time worked on clock cards to a computerized payroll system in which employees record time in and out with magnetic cards. The computer...
-
For the inertial measurement system, which testing approaches would you use? When and why?
-
The relationship between the sampling risk of incorrect acceptance and the sample size of substantive tests is a. Inverse. b. Positive. c. Indeterminate. d. None of the above.
-
form a SQL query that extracts the last 5 characters of the email address first. As you may realize, the email address always ends with '.org'. Now, how can you extract just the dot '.', from the...
-
Consider the combustion of methanol below. If 64 grams of methanol reacts with 160 grams of oxygen, what is the CHANGE in volume at STP. 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(1) The volume decreases by...
-
An ideal Brayton cycle has a net work output of 150 kJ/kg and a back work ratio of 0.4. If both the turbine and the compressor had an isentropic efficiency of 85 percent, the net work output of the...
-
During the inflation and deflation of a safety airbag in an automobile, the gas enters the airbag with a specific volume of 15 ft3/lbm and at a mass flow rate that varies with time as illustrated in...
-
Consider a U-tube whose arms are open to the atmosphere. Now water is poured into the U tube from one arm, and light oil ( = 790 kg/m3) from the other. One arm contains 70-cm-high water, while the...
-
The balance sheet of Oriental Rug Company reported the following: {Requirements} 1. Compute the book value per share for the common shares, assuming all preferred dividends are fully paid up (none in...
-
Lexington Inns reported these figures for 2020 and 2019 (in millions): {Requirement} Evaluate Lexington's return on assets and return on equity for 2020. What additional information would help you...
-
Easton Company included the following items in its financial statements for 2020, the current year (amounts in millions): Evaluate Easton's return on assets and return on equity for 2020. Payment of...

Study smarter with the SolutionInn App



