100 percent transmittance 80 60 40 20 0 2.6 2.8 3 wavelength, micrometers 3.5 4 4.5...
Fantastic news! We've Found the answer you've been seeking!
Question:
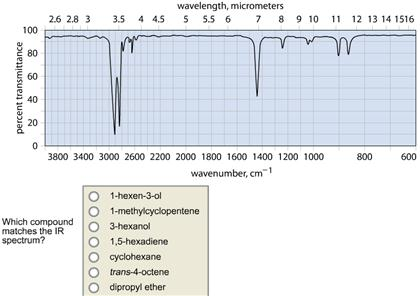
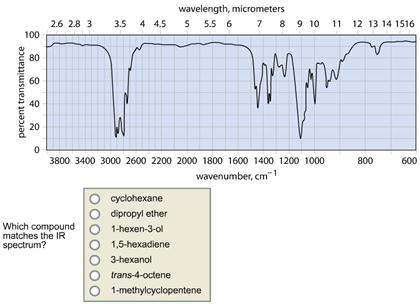
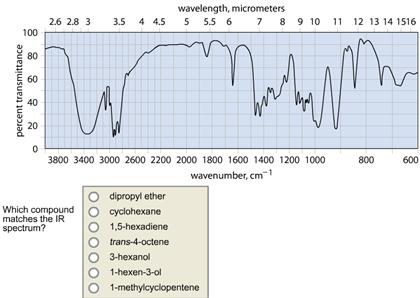
Transcribed Image Text:
100 percent transmittance 80 60 40 20 0 2.6 2.8 3 wavelength, micrometers 3.5 4 4.5 5 5.5 6 7 8 9 10 11 12 13 14 1516 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm¹ Which compound matches the IR spectrum? 1-hexen-3-ol 1-methylcyclopentene 3-hexanol 1,5-hexadiene cyclohexane trans-4-octene dipropyl ether 800 600 percent transmittance 100 80 60 40 20 0 wavelength, micrometers 2.6 2.8 3 3.5 4 4.5 5 5.5 6 7 8 9 10 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm1 Which compound matches the IR spectrum? cyclohexane dipropyl ether 1-hexen-3-ol 1,5-hexadiene 3-hexanol trans-4-octene. 1-methylcyclopentene 11 12 13 14 1516 Wh 800 600 100 percent transmittance 80 60 40 20 0 2.6 2.8 3 3.5 4 4.5 Which compound matches the IR spectrum? wavelength, micrometers 5 5.5 6 7 8 9 10 dipropyl ether cyclohexane 1,5-hexadiene trans-4-octene 3-hexanol 1-hexen-3-ol M 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm1 1-methylcyclopentene vr Mal 11 12 13 14 1516 h 800 600 100 percent transmittance 80 60 40 20 0 2.6 2.8 3 wavelength, micrometers 3.5 4 4.5 5 5.5 6 7 8 9 10 11 12 13 14 1516 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm¹ Which compound matches the IR spectrum? 1-hexen-3-ol 1-methylcyclopentene 3-hexanol 1,5-hexadiene cyclohexane trans-4-octene dipropyl ether 800 600 percent transmittance 100 80 60 40 20 0 wavelength, micrometers 2.6 2.8 3 3.5 4 4.5 5 5.5 6 7 8 9 10 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm1 Which compound matches the IR spectrum? cyclohexane dipropyl ether 1-hexen-3-ol 1,5-hexadiene 3-hexanol trans-4-octene. 1-methylcyclopentene 11 12 13 14 1516 Wh 800 600 100 percent transmittance 80 60 40 20 0 2.6 2.8 3 3.5 4 4.5 Which compound matches the IR spectrum? wavelength, micrometers 5 5.5 6 7 8 9 10 dipropyl ether cyclohexane 1,5-hexadiene trans-4-octene 3-hexanol 1-hexen-3-ol M 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm1 1-methylcyclopentene vr Mal 11 12 13 14 1516 h 800 600
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below In graph 1 we ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The cost principle states that if no cash is involved in a transaction the cash-equivalent value must be used. True False
-
3 4 5 6 7 8 9 10 11 12 13 A 14 Cost of the Asset 15 Life of the Asset in Years 16 Book Value of the Asset after 5 years 17 Depreciable Basis 18 Yearly depreciation 19 After tax Salvage Value in year...
-
3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 Mo's Motor Corp. issued $ 380,000 of 7%, 5-year bonds on April 1, 2021. Interest is payable semi-annually on April 1 and October 1. On April 1, 2023,...
-
Happy Colors manufactures crayons in a three-step process: mixing, molding, and packaging. The Mixing Department combines the direct materials of paraffin wax and pigments. The heated mixture is...
-
Dinoco Petroleum faces three potential contingency situations, described below. Dinocos fiscal year ends December 31, 2015, and it issues its 2015 financial statements on March 15, 2016. Required:...
-
What are some positive ethical efforts that are getting attention?
-
The following data are for Huang Wong Ping Retail Outlet Stores. The account balances (in thousands) are for 2017. 1. Compute (a) the cost of goods purchased and (b) the cost of goods sold. 2....
-
The following partial work sheet is taken from Kevin's Gift Shop for the year ended December 31, 20--. The ending merchandise inventory is $50,000. 1. Complete the Adjustments columns for the...
-
2. What is the computational complexity of the following function Mystery1? int Mystery1(int n) { int sum = 0; for (int i=0; i
-
A division of your company purchased a large quantity of new desktop computers during the current fiscal year. An internal audit manager has asked you to audit the process used to acquire the...
-
2. Given the following cash budget, help the firm plan for its operational cash needs by answering the questions. October November December Cash Receipts $ 37,500 15,000 $ 52.500 $ 33,750 12.500 $...
-
Craft rules for both the INPUT and OUTPUT chains to account for return traffic where necessary: Deny all traffic to and from 34.87.95.12. Deny all traffic to and from HTTP on your Linux1 server. Deny...
-
Project Aim: Overall aim of the project according to the informations below: PROPOSED CAPSTONE PROJECT Describe the problem / issue you propose to address. Community Engagement in Wind Farm...
-
Java developers need your help for below mentioned problems. Level 1: Welcome to "The Word And Number Game" Level 1: Anagram At this level, you are given a set of letters. Your task is to input all...
-
Trey Bell, president of Big Bells, Inc., strolled into his office exuding great confidence. He was convinced that his company would be able to gain a sustained competitive advantage by implementing...
-
The Australian sheep dog is a breed renowned for its intelligence and work ethic. It is estimated that 45% of adult Australian sheep dogs weigh 65 pounds or more. A sample of 14 adult dogs is...
-
For Leo-Four Plex, com ready to answer the following: 1) What controls are currently existing? 2) List out the current issues. Separate into what are problems and what are symptoms. 3) Group the...
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
When p-aminophenol reacts with one molar equivalent of acetic anhydride, a compound acetaminophen (A, C8H9NO2) is formed that dissolves in dilute NaOH. When A is treated with one equivalent of NaOH...
-
Assuming you knew the properties of the compound obtained in Problem 24.23, including its optical rotation, show how you could use periodate oxidation to distinguish methyl -D- galactopyranoside from...
-
Compound A, C6H12O2, was found to be optically active, and it was slowly oxidized to an optically active carboxylic acid B, C6H12O3, by +Ag(NH3)2. Oxidation of A by anhydrous CrO3 gave an optically...
-
The SDLC is just one model for systems development. Find at least one more and describe the differences.
-
Draw DFDs for each of these scenarios: (a) A customer goes into a bookshop and asks for this book. The member of staff looks for the book in the online stock catalogue and reports that the book is...
-
Draw an entity model to model this university scenario: A university department employs lecturers and clerical staff. It offers a three-year degree. A student has to take 12 modules during the...

Study smarter with the SolutionInn App


