12. Correct of acidic strength of the following compound is: Y H H (1) (III) CH3O...
Fantastic news! We've Found the answer you've been seeking!
Question:
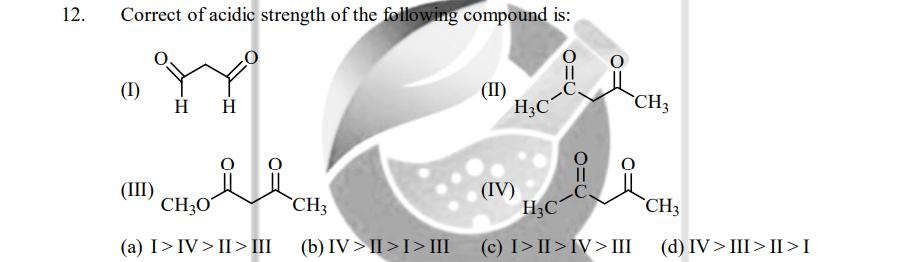
Transcribed Image Text:
12. Correct of acidic strength of the following compound is: Y H H (1) (III) CH3O CH3 (a) I>IV>II> III (b) IV>II>I> III (II) H3C (IV) =O H3C (c) I>II>IV> III CH₂ CH3 (d) IV > III>II>I 12. Correct of acidic strength of the following compound is: Y H H (1) (III) CH3O CH3 (a) I>IV>II> III (b) IV>II>I> III (II) H3C (IV) =O H3C (c) I>II>IV> III CH₂ CH3 (d) IV > III>II>I
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following compound is unstable and spontaneously decomposes to acetophenone and HBr. Give a mechanism for this transformation. OH
-
The following compound is highly activated, but nevertheless undergoes bromination very slowly. Explain.
-
The following compound is known to be chiral. Draw its enantiomer, and explain the source of chirality. CH
-
Using the information for Sarot, Inc., in SE 4 and SE 5, compute the current ratio, quick ratio, receivable turnover, days sales uncollected, inventory turnover, days inventory on hand, payables...
-
Im Really Cold Coat Company makes womens and mens coats. Both products require filler and lining material. The following planning information has been made available: Im Really Cold Coat Company does...
-
Adam, Bob, and Cathy are planning to form a partnership to create a business that will retail cell phones in a new shopping center just completed in their city. They have been able to reach agreement...
-
Problem 8 is the same as problem 7 with respect to initial measurement of the ARO liability. Now assume that Tadpoles credit standing improves over time, causing the credit-adjusted risk-free rate to...
-
Adria Lopez expects second quarter 2014 sales of her new line of computer furniture to be the same as the first quarters sales (reported below) without any changes in strategy. Monthly sales averaged...
-
Vanessa went to Florida and purchased a laptop for US$920.00 by converting his Canadian money at an exchange rate of C$1 = US$0.9254. After a month, he returned to Vancouver but the exchange rate...
-
Kidd Company produces two products. Budgeted annual income statements for the two products are provided here: Required a. Based on budgeted sales, determine the relative sales mix between the two...
-
An industrial wastewater contains 10-2 M of sodium bisulfide, NaHS, and 10-35 M of the monoprotic acid, CH5COOH. a) Determine the concentration of all species at equilibrium. b) If wastewater is to...
-
Q1: Discuss the concept of a balanced scorecard as an alternative approach to measuring business performance through key performance indicators. Q2: Explain the concept of benchmarking as a way of...
-
Consider the tri-dimensional vector channel shown below, where N is an i.i.d. normal random variable of mean 0 and variance o. N, N and N3 are independent of each other, and +02 +03-0, with 0 <02 <...
-
1. Discuss some of the interesting things you learned AND 2. Look at some of the public health policies and creative solutions to behavioral health aging problems facing our seniors. Describe what is...
-
d- Fined the optimal corner (SHI- Tomasi detector) for the following images: 1 2 3 6 7 8 11 12 13
-
Describe the four dimensions that characterize authentic leadership. Discuss how these dimensions could be incorporated in a leadership role for a type of job you are familiar with such as sales,...
-
Hutchins Company uses a predetermined overhead rate based on direct labor hours (DLH) to apply manufacturing overhead to jobs. At the beginning of the year, the company estimated manufacturing...
-
When a company has a contract involving multiple performance obligations, how must the company recognize revenue?
-
For each compound, state whether its bonding is covalent, ionic, or a mixture of covalent and ionic. (a) NaCl (b) NaOH (c) CH3Li (d) CH2CI2 (e) NaOCH3 (f) HCO2Na (g) CF4
-
Predict the products from the reactions of the following amines with sodium nitrite in dilute HCl. (a) Cyclohexanamine (b) N-ethylhexan-2-amine (c) Piperidine (d) Aniline
-
To show that (R)-2-butyl (R, R)-tartrate and (S)-2-butyl (R,R)-tartrate are not enantiomers, draw and name the mirror images of these compounds.
-
A circular shaft of radius \(r\) in the figure has a moment of inertia \(I\) and polar moment of inertia \(J\). The shaft is under torsion \(T_{z}\) in the positive zaxis and bending moment \(M_{x}\)...
-
A 20mm diameter rod made of a ductile material with a yield strength of \(350 \mathrm{MPa}\) is subject to a torque of \(T=100 \mathrm{~N} \cdot \mathrm{m}\) and a bending moment of \(M=150...
-
The figure below shows a round shaft of diameter \(1.5 \mathrm{in}\). loaded by a bending moment \(M_{z}\) \(=5,000 \mathrm{lb} \cdot\) in., a torque \(T=8,000 \mathrm{lb} . \cdot\) in., and an axial...

Study smarter with the SolutionInn App



