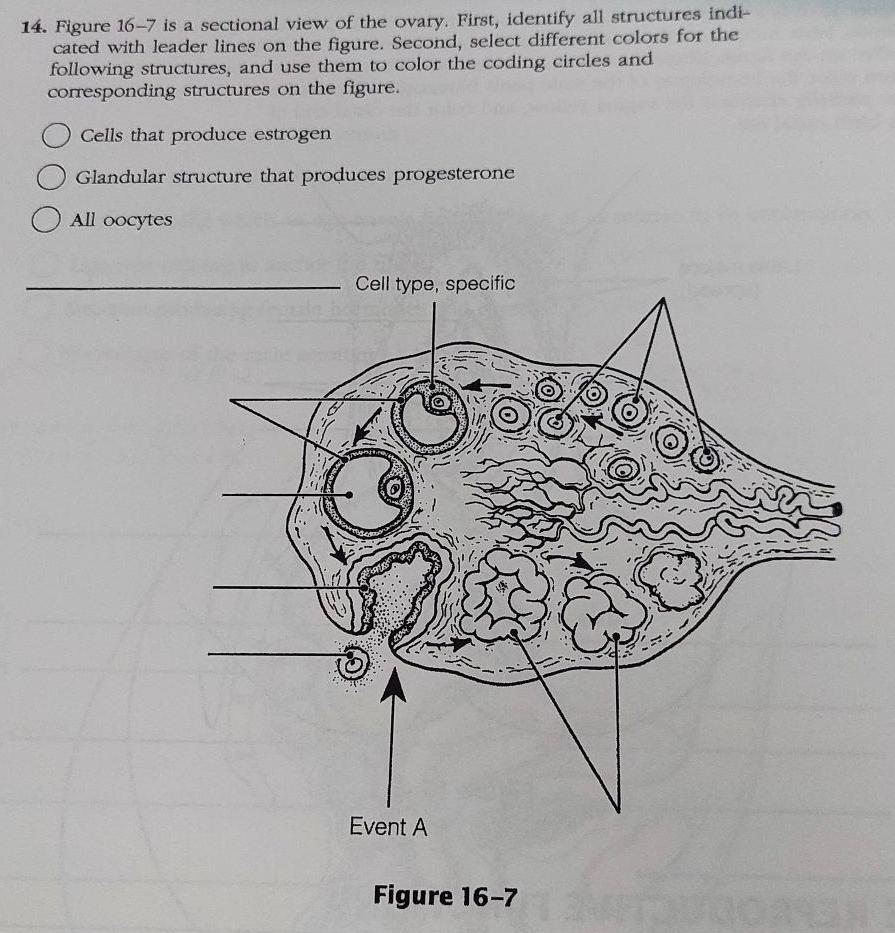
14. Figure 16-7 is a sectional view of the ovary. First, identify all structures indi- cated...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
14. Figure 16-7 is a sectional view of the ovary. First, identify all structures indi- cated with leader lines on the figure. Second, select different colors for the following structures, and use them to color the coding circles and corresponding structures on the figure. Cells that produce estrogen Glandular structure that produces progesterone O All oocytes Cell type, specific Event A Figure 16-7 14. Figure 16-7 is a sectional view of the ovary. First, identify all structures indi- cated with leader lines on the figure. Second, select different colors for the following structures, and use them to color the coding circles and corresponding structures on the figure. Cells that produce estrogen Glandular structure that produces progesterone O All oocytes Cell type, specific Event A Figure 16-7
Expert Answer:
Answer rating: 100% (QA)
14 Figure 167 is a sectional view of the ovary First identify ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Helmboltz Coils. Fig. is a sectional view of two circular coils with radius a, each wound with N turns of wire carrying a current I, circulating in the same direction in both coils. The coils are...
-
Copy the vectors in the figure and use them to draw each of the following vectors. (a) a + b (b) a - b (c) -1/2a (d) 2a + b a
-
Choose two rays in Fig. 23.7 and use them to prove that the angle of incidence is equal to the angle of reflection Reflected rays Incident rays Reflected wavefront Incident wavefronts Wavelet
-
5. Explain how Culture can help companies set legal and ethical standards?
-
Refer to the data on sanitation levels of cruise ships, presented in Exercise. These data are saved in the SHIPSANIT file. a. Use the box plot method to detect any outliers in the data. b. Use the...
-
Ten items that may or may not be involved in the bank reconciliation process for April are listed in the table shown below: Instructions Complete the table shown above, identifying where each item...
-
Describe the various forms of sterilization.
-
Sunco processes oil into aviation fuel and heating oil. It costs $40 to purchase each 1000 barrels of oil, which is then distilled and yields 500 barrels of aviation fuel and 500 barrels of heating...
-
Use the transaction information provided for Mike's Motors (the previous question) to create a balance sheet for Mike's Motors. Make sure to include every account that you used in the journal entries...
-
Wilhelmina is a trader whose financial year ends on 31 March. Her trial balance at 31 March 2004 was as follows. Further information 1. Stock at 31 March 2004 cost $16.000. 2. The loan was received...
-
How many unique processes and threads are created in the following program segment? pid_t pid; pid = fork(); if (pid == 0) { /* child process */ fork(); thread.create(); } else { /* parent process */...
-
A company provided the following information: Standard fixed overhead rate (SFOR) per direct labour hour $7.00 Actual fixed overhead rate (AFOR) per direct labour hour $6.95 Actual direct labour...
-
On January 1, 2024, a company began construction of an automated cattle feeder system. The system was finished and ready for use on September 30, 2025. Expenditures on the project were as follows:...
-
For the function f(x) = 2x ln x, find f'(x). f'(x) = Preview
-
Find f'(x) for f(x) = ln(2x - 2x + 2). f'(x) = Preview
-
Find f'(x) for f(x) = ln((x + 3)). f'(x) = Preview TIP
-
The following details have been provided by Osheaga Ltd. for the year ended December 31, 2023. Osheaga Ltd. Income Statement For the Year Ended December 31, 2023 Sales revenue Cost of goods sold...
-
You are thinking of investing in one of two companies. In one annual report, the auditors opinion states that the financial statements were prepared in accordance with generally accepted accounting...
-
A certain oxide of titanium is 28.31% oxygen by mass and contains a mixture of Ti2+ and Ti3+ ions. Determine the formula of the compound and the relative numbers of Ti2+ and Ti3+ ions.
-
Differentiate between what happens when the following are dissolved in water. a. Polar solute versus nonpolar solute b. KF versus C6H12O6 c. RbCl versus AgCl d. HNO3 versus CO
-
Give the name and formula of the binary compound formed by each of the following pairs of elements. a. Li and N b. Na and Br c. K and S d. Li and P e. Rb and H f. Na and H
-
Who was Phar-Mors flamboyant Chief Executive Officer?
-
Which of the following generally is not considered something of value? 1. Cash, money or checks 2. Airline miles or hotel credits associated with frequent activity (e.g., frequent flier miles) 3. An...
-
Which of the following is not one of the five major categories of fraudulent disbursements? 1. Payroll schemes 2. Expense reimbursement schemes 3. Shell company schemes 4. Billing schemes

Study smarter with the SolutionInn App


