14) What element is undergoing oxidation (if any) in the following reaction? Zn(s) + 2 AgNO3(aq)...
Fantastic news! We've Found the answer you've been seeking!
Question:
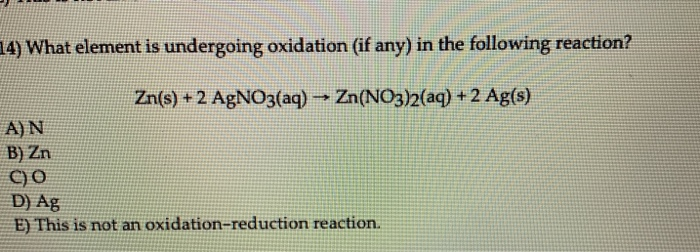
Transcribed Image Text:
14) What element is undergoing oxidation (if any) in the following reaction? Zn(s) + 2 AgNO3(aq) → Zn(NO3)2(aq) + 2 Ag(s) A) N B) Zn C)O D) Ag E) This is not an oxidation-reduction reaction. 14) What element is undergoing oxidation (if any) in the following reaction? Zn(s) + 2 AgNO3(aq) → Zn(NO3)2(aq) + 2 Ag(s) A) N B) Zn C)O D) Ag E) This is not an oxidation-reduction reaction.
Expert Answer:
Answer rating: 100% (QA)
Zn is undergoing oxidation in this reaction in an oxida... View the full answer

Related Book For 

Organic Chemistry A Short Course
ISBN: 978-1111425562
13th edition
Authors: Harold Hart, Christopher M. Hadad, Leslie E. Craine, David J. Hart
Posted Date:
Students also viewed these chemistry questions
-
In the following reaction of trichloroacetic acid, HC 2 Cl 3 O 2 , with formate ion, CHO 2 , the formation of trichloroacetate ion, C 2 Cl 3 O 2 , and formic acid, HCHO 2 is favored. Which is the...
-
Which atoms in the following reaction undergo changes in their oxidation state? Which atom is oxidized? Which one is reduced? 2CH3CH2OH + 2Na 2CH3CH2ONa + H2
-
Predict the product(s) formed in the following reaction and explain fully the reaction mechanism (where stereochemical issues are involved you will need to indicate the precise stereochemical...
-
If the demand function and supply function for Z-brand phones are p + 2q = 100 and 35p + 20q = 350, respectively, compare the quantity demanded and the quantity supplied when p = 14. Are there...
-
The Aztec Company stocks a variety of parts and materials it uses in its manufacturing processes. Recently, as demand for its finished goods has increased, management has had difficulty managing...
-
Shetland Inc. had pretax financial income of $154,000 in 2020. Included in the computation of that amount is insurance expense of $4,000 which is not deductible for tax purposes. In addition,...
-
What are the different ways a deposition summary may be prepared?
-
Peter and Tanya Rothing operated Diamond R Stables near Belgrade, Montana, where they bred, trained, and sold horses. Arnold Kallestad owned a ranch in Gallatin County, Montana, where he grew hay and...
-
From these websites: https://writingcommons.org/section/mindset/ https://www.psychologytoday.com/us/blog/click-here-happiness/201904/15-ways-build-growth-mindset What strikes you as most valuable in...
-
Tikkity Tock Clock Company sells three types of clocks. They purchase these clocks from manufacturers and sell them to retail stores. Tikkity Tock uses a perpetual inventory system, weighted average...
-
1 . calculate the amount of joint costs that should be assigned to the main products using the Net Realizable Value Method for allocation of joint costs. All calculations need to be done using formul...
-
In 1970, Italian Gianluigi Aponte founded the Mediterranean Shipping Company (MSC) with a single second-hand ship and just $5,000 in capital to move cargo between Europe and Africa. Despite a...
-
Beverage Giant Coca-Cola faces several challenges as a result of an increase of consumer health awareness, the reduction of carbonated drinks in schools in many regions, and newer, healthier drinks....
-
Companies large and small, across all industries, are crowdsourcing product innovation ideas. Research three crowdsourcing campaigns that companies have used within the past two years. Were they...
-
Once upon a time, there were two major forces dominating the discount retail market: Walmart and Target. The two retailers offered very similar merchandise assortments, and their stores were close to...
-
According to the U.K. price comparison site Compare the Market, the cost of funerals is rising on average at a rate of 7 percent per year. The Funeral Planning Authority (FPA) estimates that the...
-
What is the main advantage of a 'Multinational Corporation' ( ( MNC ) ? ) ? a ) ) Uniformity in marketing practices across different countries b ) ) Reduction in operational costs due to global...
-
1. Which of the four major types of information systems do you think is the most valuable to an organization? 2. How do you critically associate the ideas of business agility and business efficiency...
-
Cholesterol has a rigid fused-ring structure (page 454) and is found in phospholipid bilayers where it adds rigidity to cell membranes. Cholesterol contains both polar and non polar groups. Taking...
-
Which of the following substances contain stereogenic centers? a. 2,2-dibromobutane b. 3-methylcyclopentene c. 1,2-difluoropropane d. 2,3-dimethylheptane e. methylcyclobutane f. 1-deuteriopropanol...
-
Compare the intermediate benzenonium ions for ortho, meta and para bromination of benzoic acid, and explain why the main product is m-bromobenzoic acid. C-OH benzoic aciod
-
Determine the greatest magnitude of the applied forces P if the allowable bending stress is ( c ) allow = 50 MPa in compression and ( t ) allow =120 MPa in tension. 250 mm 75 mm 10 mm 10 mm- 150 mm...
-
If P = 6kN, determine the maximum tensile and compressive bending stresses in the beam. 250 mm 75 mm 10 mm 10 mm- 150 mm 160 mm 10 mm P 150 mm
-
The bar is subjected to a moment M = 40 Nm of Determine the smallest radius r of the fillets so that an allowable bending stress of allow = 124MPa is not exceeded. M 20 mm 80 mm 7 mm M

Study smarter with the SolutionInn App


