16.5 g of CO are dissolved in 500 mL of water, which has density 1.00 kg...
Fantastic news! We've Found the answer you've been seeking!
Question:
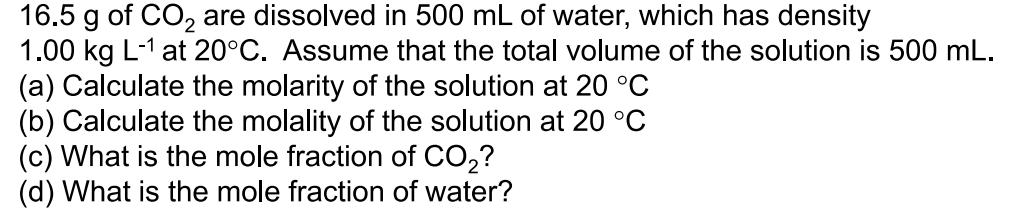
Transcribed Image Text:
16.5 g of CO are dissolved in 500 mL of water, which has density 1.00 kg L-1 at 20C. Assume that the total volume of the solution is 500 mL. (a) Calculate the molarity of the solution at 20 C (b) Calculate the molality of the solution at 20 C (c) What is the mole fraction of CO? (d) What is the mole fraction of water? 16.5 g of CO are dissolved in 500 mL of water, which has density 1.00 kg L-1 at 20C. Assume that the total volume of the solution is 500 mL. (a) Calculate the molarity of the solution at 20 C (b) Calculate the molality of the solution at 20 C (c) What is the mole fraction of CO? (d) What is the mole fraction of water?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
3.50 grams of glycine are dissolved in 500 mL of distilled water. a) How many milliliters of 1.0 M NaOH are needed to bring the pH to 10.15? b)How many milliliters of 0.25 M HCl are needed to bring...
-
Assume that the total sales value at the split-off point for product 1 is $50,000 instead of $130,000 and the sales value of product 3 is $2,000 instead of $20,000. Assume also that, because of its...
-
Assume that the total sales value at the split-off point for product 1 is $50,000 instead of $130,000 and the sales value of product 3 is $2,000 instead of $20,000. Assume also that, because of its...
-
C.J mensah started a new park business called Jane park on February 1, 2016. The following selected events and transaction occurs during February. February 1. Invested $40,000 by issuing a common...
-
Young Software began with cash of $13,000. Young then bought supplies for $1,800 on account. Separately, Young paid $4,000 for a computer. Answer these questions. a. How much in total assets does...
-
On March 1, 2019, Elkhart enters into a new contract to build a specialized warehouse for $7 million. The promise to transfer the warehouse is determined to be a performance obligation. The contract...
-
Integral balances can also be used for heat transfer in a turbulent-flow boundary layer if a form for the velocity profile is assumed. A common form is the 1/7th-power law:...
-
Given that z is a standard normal random variable, compute the following probabilities. a. P(z 1.0) b. P(z 1) c. P(z 1.5) d. P(2.5 z) e. P( 3 < z 0)
-
A lake with a fixed carrying capacity contains a certain fish population. The fish population in the lake has a growth rate that is proportional to its size when the population is very small relative...
-
Hong Kong Island features steep, hilly terrain, as well as hot and humid weather. Travelling up and down the slopes therefore causes problems; this has led the city authorities to imagine rather...
-
A409 - Tax Return #1 - Spring 2024 Instructions: 2023 return with the information below. I need help filling out Forms 1040, 8812, two 8814s (one for Ariana and one for Steven), and Schedules 1, 2,...
-
To construct a confidence interval for each of the following quantities, say whether it would be better to use paired samples or independent samples. a. The mean difference in weight loss between...
-
Scores on a certain IQ test are known to have a mean of 100 . A random sample of 60 students attend a series of coaching classes before taking the test. Let \(\mu\) be the population mean IQ score...
-
A sample of size \(n=100\) is used to test \(H_{0}: \mu \leq 20\) versus \(H_{1}: \mu>20\). The value of \(\mu\) will not have practical significance unless \(\mu>25\). The population standard...
-
The rejection region for a \(5 \%\) level test of \(H_{0}: \mu \geq 10\) versus \(H_{1}: \mu <10\) is \(\bar{X} <7.9\). Find the rejection region for a \(1 \%\) level test.
-
A process that manufactures steel bolts is supposed to be calibrated to produce bolts with a mean length of \(5 \mathrm{~cm}\). A sample of 100 bolts has a mean length of \(5.02 \mathrm{~cm}\). The...
-
Describe what elasticity of demand measures and the three possible variations. Describe the effect of a lower price on total revenue when demand is elastic, when demand is inelastic and when demand...
-
An educational researcher devised a wooden toy assembly project to test learning in 6-year-olds. The time in seconds to assemble the project was noted, and the toy was disassembled out of the childs...
-
Use the Born-Haber cycle outlined in Section 9.3 for LiF to calculate the lattice energy of NaCl. [The heat of sublimation of Na is 108 kJ/mol and Hf (NaCl) = - 411 kJ/mol. Energy needed to...
-
The He+ ion contains only one electron and is therefore a hydrogenlike ion. Calculate the wavelengths, in increasing order, of the first four transitions in the Balmer series of the He+ ion. Compare...
-
Fishes in the Antarctic Ocean swim in water at about - 22C. (a) To prevent their blood from freezing, what must be the concentration (in molality) of the blood? Is this a reasonable physiological...
-
Select a publicly traded company or use a company assigned by your instructor. Using the firms most recent Form 10-K report (accessed through the SEC EDGAR database or from the companys website),...
-
Select a publicly traded company for analysis or use a company assigned by your instructor. Using the firms most recent Form 10-K report (accessed through the SEC EDGAR database or from the companys...
-
Select a publicly traded company for analysis. Using the firms most recent 10-K Report (accessed through the SEC EDGAR database at sec.gov or from the companys website), identify how financial data...

Study smarter with the SolutionInn App



