1.This experiment was performed and 10.56 grams of distillatewas obtained. The density of the distillate at 25?C...
Question:
1.This experiment was performed and 10.56 grams of distillatewas obtained. The density of the distillate at 25?C wasdetermined to be 0.814 g/mL. Use the below to calculatethe mass of ethanol, in grams, obtained in the experiment. Showyour calculations.
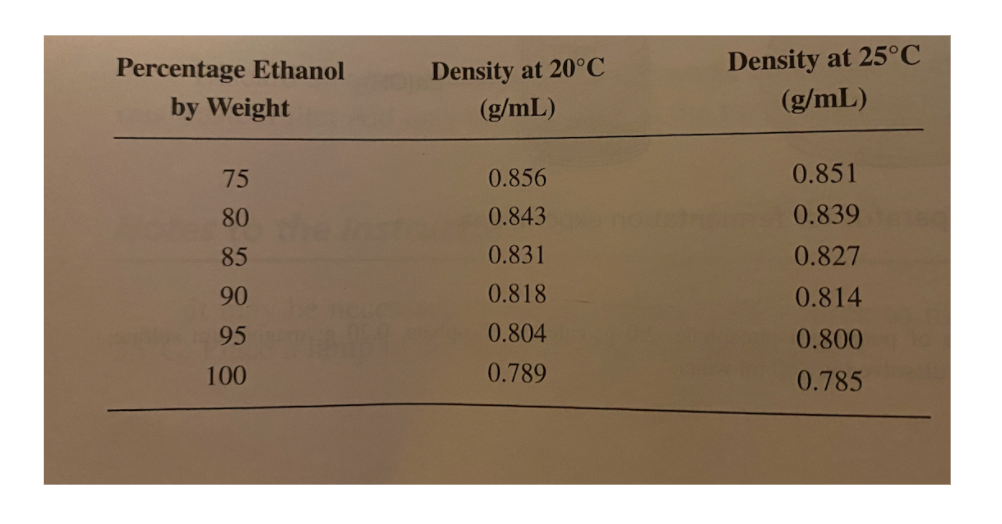
2. The total volume of fermentation liquid is 200 mL but thelargest distillation flask available for today?s lab will only holda total volume of 50 mL. Instead of performing thedistillation multiple times, we will perform the distillation onlyonce using a representative sample of the fermentation liquid. Ifyou use 40 mL of the fermentation liquid as your representativesample and obtain 1.5 g of ethanol, what mass of ethanol should youuse as your actual yield inyour percent yield calculations?

Introduction to Statistical Quality Control
ISBN: 978-1118146811
7th edition
Authors: Douglas C Montgomery




