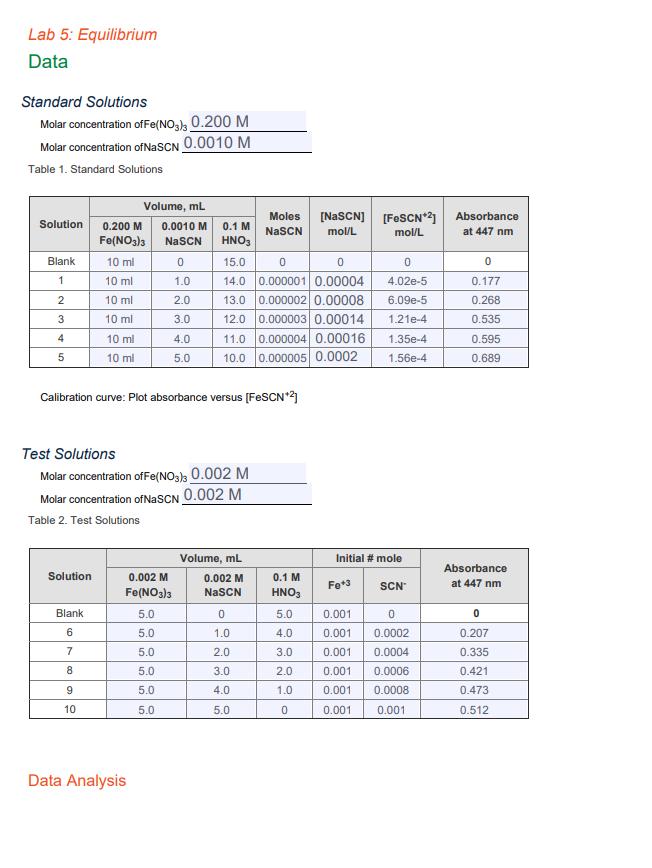
Lab 5: Equilibrium Data Standard Solutions Molar concentration of Fe(NO3)3 0.200 M Molar concentration of NaSCN...
Fantastic news! We've Found the answer you've been seeking!
Question:
![Table 3: Equilibrium Conditions Using theequation ( K_{e q}=frac{left[mathrm{FeSCN}^{+2}ight]}{left[F e^{+3} mathrm{I](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/08/62e9062cb9e05_1659438634771.jpg)
Transcribed Image Text:
Lab 5: Equilibrium Data Standard Solutions Molar concentration of Fe(NO3)3 0.200 M Molar concentration of NaSCN 0.0010 M Table 1. Standard Solutions Solution 0.200 M Fe(NO3)3 10 ml 10 ml 10 ml 10 ml Blank 1 2 3 4 5 Solution Blank 6 10 ml 10 ml 7 8 Volume, mL 0.0010 M NaSCN 0 1.0 2.0 3.0 4.0 5.0 9 10 Calibration curve: Plot absorbance versus [FeSCN*2] Test Solutions Molar concentration of Fe(NO3)3 0.002 M Molar concentration of NaSCN 0.002 M Table 2. Test Solutions 0.1 M HNO3 0.002 M Fe(NO3)3 5.0 5.0 5.0 5.0 5.0 5.0 Data Analysis 15.0 0 0 14.0 0.000001 0.00004 13.0 0.000002 0.00008 12.0 0.000003 0.00014 Moles NaSC 11.0 0.000004 0.00016 1.35e-4 10.0 0.000005 0.0002 1.56e-4 Volume, mL 0.002 M NaSCN 0 1.0 2.0 3.0 4.0 5.0 [NaSCN] [FeSCN*2] Absorbance mol/L mol/L at 447 nm 0.1 M HNO3 5.0 4.0 3.0 2.0 1.0 0 0 4.02e-5 6.09e-5 1.21e-4 Initial # mole Fe+3 0.001 0.001 0.001 0.001 0.001 0.001 SCN 0 0.0002 0.0004 0.0006 0.0008 0.001 0 0.177 0.268 0.535 0.595 0.689 Absorbance at 447 nm 0 0.207 0.335 0.421 0.473 0.512 Table 3: Equilibrium Conditions Solution 6 7 8 9 10 Solution 6 7 8 9 10 [FeSCN¹21" mol/L [FeSCN*2]** mol/L Iron Equilibrium Moles Moles Fe*3 FeSCN¹2 reacted Using the equation K = Solution 6 Keq = Solution 7 Keq = Solution 8 Keq= Solution 9 Keq = Solution 10 Keq = Ave Keq = [FeSCN+2] eq [Fe +³[SCN] Moles Fe*3 unreacted [Fe*3] unreacted, mol/L Thiocyanate Equilibrium Moles Moles SCN Moles SCN [SCN] unreacted, SCN reacted unreacted mol/L calculate the equilibrium constant for solution 6 below: Lab 5: Equilibrium Data Standard Solutions Molar concentration of Fe(NO3)3 0.200 M Molar concentration of NaSCN 0.0010 M Table 1. Standard Solutions Solution 0.200 M Fe(NO3)3 10 ml 10 ml 10 ml 10 ml Blank 1 2 3 4 5 Solution Blank 6 10 ml 10 ml 7 8 Volume, mL 0.0010 M NaSCN 0 1.0 2.0 3.0 4.0 5.0 9 10 Calibration curve: Plot absorbance versus [FeSCN*2] Test Solutions Molar concentration of Fe(NO3)3 0.002 M Molar concentration of NaSCN 0.002 M Table 2. Test Solutions 0.1 M HNO3 0.002 M Fe(NO3)3 5.0 5.0 5.0 5.0 5.0 5.0 Data Analysis 15.0 0 0 14.0 0.000001 0.00004 13.0 0.000002 0.00008 12.0 0.000003 0.00014 Moles NaSC 11.0 0.000004 0.00016 1.35e-4 10.0 0.000005 0.0002 1.56e-4 Volume, mL 0.002 M NaSCN 0 1.0 2.0 3.0 4.0 5.0 [NaSCN] [FeSCN*2] Absorbance mol/L mol/L at 447 nm 0.1 M HNO3 5.0 4.0 3.0 2.0 1.0 0 0 4.02e-5 6.09e-5 1.21e-4 Initial # mole Fe+3 0.001 0.001 0.001 0.001 0.001 0.001 SCN 0 0.0002 0.0004 0.0006 0.0008 0.001 0 0.177 0.268 0.535 0.595 0.689 Absorbance at 447 nm 0 0.207 0.335 0.421 0.473 0.512 Table 3: Equilibrium Conditions Solution 6 7 8 9 10 Solution 6 7 8 9 10 [FeSCN¹21" mol/L [FeSCN*2]** mol/L Iron Equilibrium Moles Moles Fe*3 FeSCN¹2 reacted Using the equation K = Solution 6 Keq = Solution 7 Keq = Solution 8 Keq= Solution 9 Keq = Solution 10 Keq = Ave Keq = [FeSCN+2] eq [Fe +³[SCN] Moles Fe*3 unreacted [Fe*3] unreacted, mol/L Thiocyanate Equilibrium Moles Moles SCN Moles SCN [SCN] unreacted, SCN reacted unreacted mol/L calculate the equilibrium constant for solution 6 below:
Expert Answer:
Answer rating: 100% (QA)
Solution Since So Solution 1 2 345 02 M fet3 10 Solution 1 Nasen is ... View the full answer

Related Book For 

Financial Reporting and Analysis Using Financial Accounting Information
ISBN: 978-1439080603
12th Edition
Authors: Charles H Gibson
Posted Date:
Students also viewed these chemistry questions
-
J Meyers has the following budgeted figures for the year ended 30 June 2021: Units produced Machine-hours Direct labor-hours Manufacturing overheads Direct materials Direct wages 194 400 7 920 13 440...
-
General Long-term Liability and Capital Asset Transactions The Village of Nassau issued a 3-year, 6 percent note in the amount of $100,000 to finance the purchase of vehicles for the Public Works...
-
Capital Leases that have not been capitalized will decrease the times interest earned ratio. Comment.
-
Explain why each of the following statements about profit-maximizing competitive firms is incorrect. Restate each one correctly. a. A competitive firm will produce output up to the point where price...
-
1) What does Coca-Cola stand for? Is it the same for everyone? Explain. 2) Coca-Cola has successfully marketed to billions of people around the world. Why is it so successful? 3) Can Pepsi or any...
-
A circuit system is given in Figure. Assume the components fail independently.(a) What is the probability that the entire system works?(b) Given that the system works, what is the probability that...
-
Find the z-score that has 43.5% of the distributions area to its left. Find the indicated z-score.
-
A certain industrial firm desires an economic analysis to determine which of two different machines should be purchased. Each machine is capable of performing the same task in a given amount of time....
-
Acme Corp. just paid a dividend of $3.00 per share (ie., D 0 = $3.00). The dividend is expected to grow at a constant rate of 4% per year. What is the expected dividend at the end of year three (D 3...
-
Martin Manufacturing is preparing its master budget for the first quarter of the upcoming year. The following data pertain to Martin Manufacturing's operations: Current Assets as of December 31...
-
Answer the following 3 questions (that build on your previous Starbucks assignment) with at least *750* words in your original post. You cannot access any other responses until you have submitted...
-
Use the product, quotient, and power rules, as necessary, to simplify the following expression. Write the answer with only positive exponents. (810-2)4 (4103)4 The answer is (810-2)4 (4103)4...
-
Write a Java program to sort an array of strings in alphabetical order using the bubble sort algorithm.
-
Consider a variation on the Hay Farm problem where Cassidy grows Alfalfa and Orchard grass. Cassidy can rent the cutter for up to 21 hours, the rake for up to r hours, and the baler for up to 16...
-
Using a multi-business organization of your choice in Zambia, illustrate the concept of SBU. Discuss the levels of business strategy you have identified and demonstrate how goals and objectives vary...
-
Write a depth-first search (DFS) algorithm in Java to traverse a directed graph represented as an adjacency list.
-
How do protagonists in Bildungsroman and coming-of-age narratives undergo psychological and moral growth, grappling with the challenges of self-discovery, autonomy, and individuation as they...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Indicate some sources that contain an appraisal of the outlook for particular industries.
-
The following material relates to Darrow Company: Required Place an X in the appropriate columns for each of thesituations. Cash Flows Classification Noncash Effect on Cash Operating Investing...
-
Hess Corporation* included the information in this case as part of the supplementary oil and gas data. This case only represents a small portion of the supplementary oil and gas data. Results of...
-
While the BohrSommerfeld condition sometimes gets the energy eigenvalues exactly correct, it can also be used for systems where the exact solution is not known. In this example, we will estimate the...
-
It's useful to see how our quantum perturbation theory works in a case that we can solve exactly. Let's consider a two-state system in which the Hamiltonian is...
-
Let's see how the variational method works in another application. Let's assume we didn't know the ground-state energy of the quantum harmonic oscillator and use the variational method to determine...

Study smarter with the SolutionInn App


