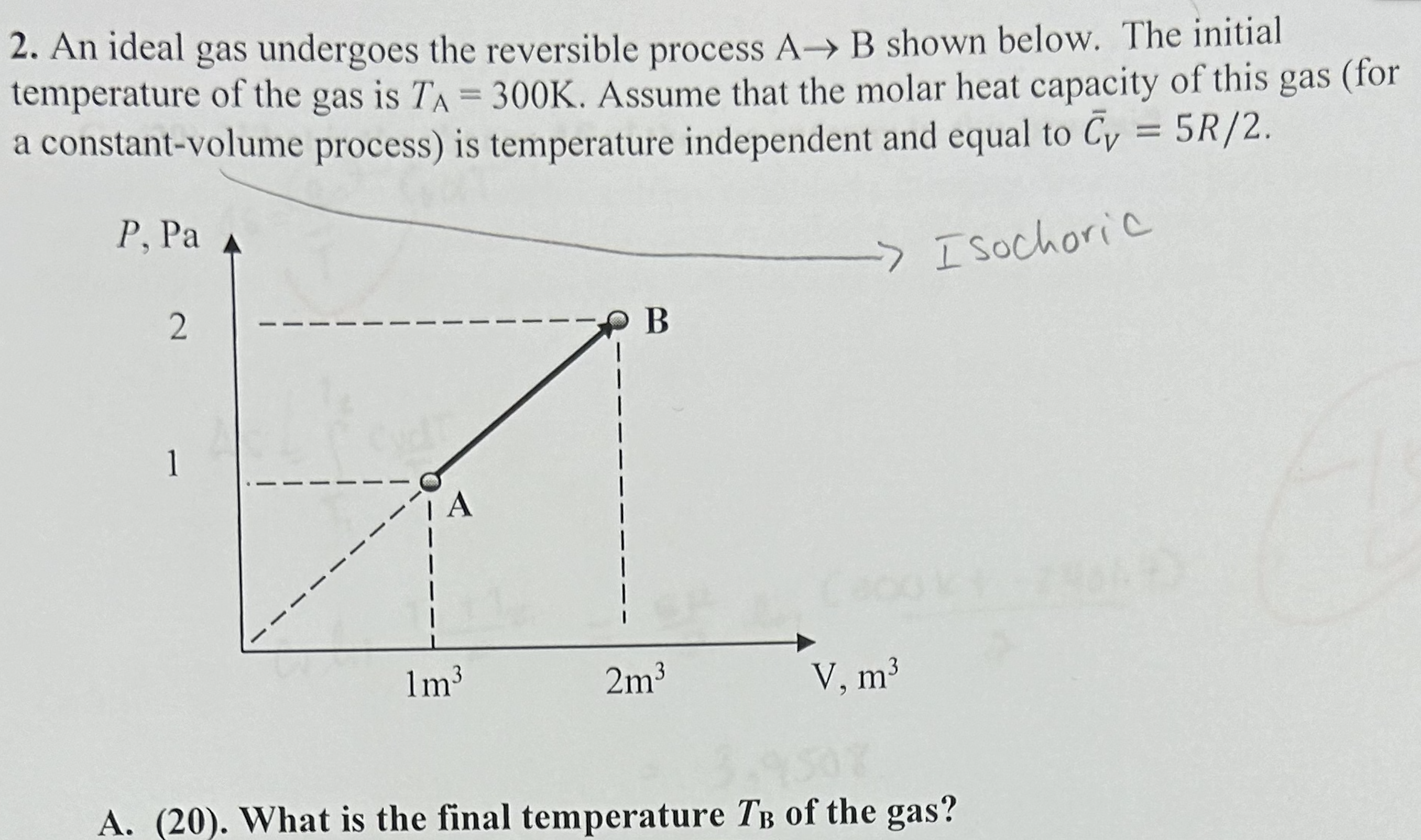
2. An ideal gas undergoes the reversible process A B shown below. The initial temperature of...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
2. An ideal gas undergoes the reversible process A B shown below. The initial temperature of the gas is TA = 300K. Assume that the molar heat capacity of this gas (for a constant-volume process) is temperature independent and equal to Cy = 5R/2. P, Pa 2 -> Isochoric B 1 1m 2m V, m 9507 A. (20). What is the final temperature TB of the gas? 4. A vessel contains n moles of an ideal gas 1 and 2 moles of ideal gas 2 separated by a wall. Both gases are at the same pressure P and temperature T equal to the temperature of the surroundings. The vessel is not thermally insulated from the surroundings. T n2 P, T P, T A. (35) Calculate the change AS of the entropy of the gases after the wall is removed and the gases are mixed. 3(50). A cup of hot coffee at T = 360K is brought to the classroom, whose temperature is T = 300K. The heat capacity of the coffee in the cup is Cp = 400J/K, and the heat capacity of the cup itself is negligible. Estimate the change of the entropy of the Universe resulting from the coffee getting cold and attaining the room temperature. For the purpose of this problem the Universe = coffee + surroundings, where you can think of the surroundings as a giant body (of infinitely large heat capacity) at T = T. 2. 5* (70 extra credit points). A balloon filled with hot air at 7=400K and P = 1 atm is released at sea level and rises until it stops h meters above sea level. The balloon is made from a weightless, rigid material that does not conduct heat such that the volume of the balloon remains constant, and there is no the heat exchange between the balloon and the surroundings. The atmosphere is an ideal gas whose temperature is constant and equal to T = 300K at all elevations. Also assume that the atmospheric pressure changes with the elevation h above sea level according to the barometric formula (derived in the class): P=P exp(-Mgh/RT), where Po= 1 atm and the average molar mass of air is M = 29 g/mol. (Yes, this is the balloon from HW1). Estimate the change of the entropy of the surroundings (i.e., the atmosphere) as a result of this process (i.e. after the balloon stops rising). 2. An ideal gas undergoes the reversible process A B shown below. The initial temperature of the gas is TA = 300K. Assume that the molar heat capacity of this gas (for a constant-volume process) is temperature independent and equal to Cy = 5R/2. P, Pa 2 -> Isochoric B 1 1m 2m V, m 9507 A. (20). What is the final temperature TB of the gas? 4. A vessel contains n moles of an ideal gas 1 and 2 moles of ideal gas 2 separated by a wall. Both gases are at the same pressure P and temperature T equal to the temperature of the surroundings. The vessel is not thermally insulated from the surroundings. T n2 P, T P, T A. (35) Calculate the change AS of the entropy of the gases after the wall is removed and the gases are mixed. 3(50). A cup of hot coffee at T = 360K is brought to the classroom, whose temperature is T = 300K. The heat capacity of the coffee in the cup is Cp = 400J/K, and the heat capacity of the cup itself is negligible. Estimate the change of the entropy of the Universe resulting from the coffee getting cold and attaining the room temperature. For the purpose of this problem the Universe = coffee + surroundings, where you can think of the surroundings as a giant body (of infinitely large heat capacity) at T = T. 2. 5* (70 extra credit points). A balloon filled with hot air at 7=400K and P = 1 atm is released at sea level and rises until it stops h meters above sea level. The balloon is made from a weightless, rigid material that does not conduct heat such that the volume of the balloon remains constant, and there is no the heat exchange between the balloon and the surroundings. The atmosphere is an ideal gas whose temperature is constant and equal to T = 300K at all elevations. Also assume that the atmospheric pressure changes with the elevation h above sea level according to the barometric formula (derived in the class): P=P exp(-Mgh/RT), where Po= 1 atm and the average molar mass of air is M = 29 g/mol. (Yes, this is the balloon from HW1). Estimate the change of the entropy of the surroundings (i.e., the atmosphere) as a result of this process (i.e. after the balloon stops rising).
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
You are considering a job offer. The job offers an annual salary of $42,000, $45,000, and $48,000 a year for the next three years, respectively. The offer also includes a starting bonus of $1,000...
-
You have been appointed a project manager for a small software products company. Your job is to build a breakthrough product that combines virtual reality hardware with state-of-the-art software....
-
Without using calculus (as in the previous exercise), show that, if n is a power of 2 greater than 1, then, for H n , the nth harmonic number, H n 1 + H n/2 Use this fact to conclude that Hn 1 +...
-
What are the different types of schemes associated with complex frauds?
-
Presented below is financial information for two different companies. Instructions (a) Determine the missing amounts. (b) Determine the gross profit rates. (Round to one decimalplace.) Lee Company n...
-
B: Point out the error in the code and correct it. class Person { public void int display () { System.out.println("This is a Person"); } } class Student extnd Person { public void display () {...
-
SureTight Company began work on a four year construction project on January 1, 2007 and uses the percentage of completion method. The contract price was $4,000,000 and estimated total costs were...
-
Y Corp. issued bonds on May 1 of the current year. These bonds have interest payment dates of May 1 and November 1 of each year. The bond interest expense account for the calendar year ended December...
-
1- What do you understand by common law? 2. What are some advantages and disadvantages of the common law system?
-
1. A cannonball is fired from a height of 30 m above the ground with an initial velocity of 40 m/s at an angle of 30 above the horizontal. a. Find the time of flight. b. Determine the horizontal...
-
Samantha and Diane are a same sex couple who have been living together for fifteen years, who never intended to be married as they were just fine cohabitating for the duration of their lives. They...
-
Is the United States a common law or civil jurisdiction? What are some key differences between common law and civil law jurisdictions (please state at least two key differences)? In international...
-
A ten-year bond has a face value of $10,000, a face interest rate of 11 percent, an unamortized bond premium of $400, and an effective interest rate of 10 percent. In the first semiannual interest...
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
As the final edits to the Sixth Edition were being made, the coronavirus pandemic struck and it was decided to expand Problem P9-7 and apply it to the pandemic. (Mayur Tikmani, Devosmita Sen, Manjeet...
-
Styrene can be produced from ethylbenzene by the following reaction: ethylbenzenestyrene+H2(1) However, several irreversible side reactions also occur: ethylbenzene benzene + ethylene (2)...
-
The elementary gas-phase reaction A + B 2C in Problem P11-8B is now continued and carried out in packed-bed reactor. The entering molar flow rates are F A0 = 5 mol/s, F B0 = 2F A0 , and F I = 2F A0...
-
Why has subordinate legislation come to prominence?
-
What are the advantages of a settlement over a court trial?
-
Define appellant ; respondent ; counterclaim ; counsel ; bench ; writ ; settlement ; pleadings ; party and party costs ; res judicata .

Study smarter with the SolutionInn App


