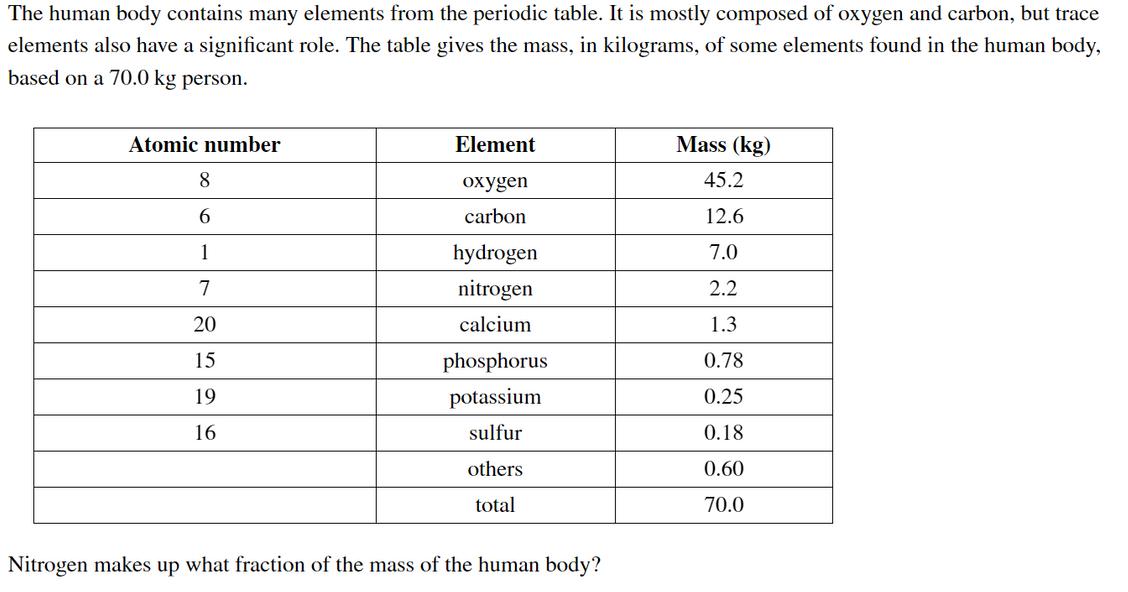
The human body contains many elements from the periodic table. It is mostly composed of oxygen...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
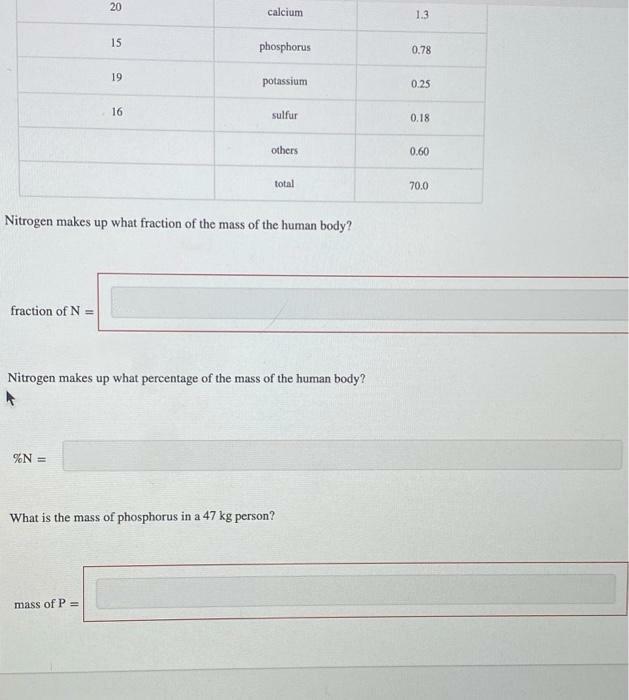
The human body contains many elements from the periodic table. It is mostly composed of oxygen and carbon, but trace elements also have a significant role. The table gives the mass, in kilograms, of some elements found in the human body, based on a 70.0 kg person. Atomic number Element Mass (kg) 8 oxygen 45.2 6. carbon 12.6 1 hydrogen 7.0 7 nitrogen 2.2 20 calcium 1.3 15 phosphorus 0.78 19 potassium 0.25 16 sulfur 0.18 others 0.60 total 70.0 Nitrogen makes up what fraction of the mass of the human body? 20 calcium 1.3 15 phosphorus 0.78 19 potassium 0.25 16 sulfur 0.18 others 0.60 total 70.0 Nitrogen makes up what fraction of the mass of the human body? fraction of N = Nitrogen makes up what percentage of the mass of the human body? %N = What is the mass of phosphorus in a 47 kg person? mass of P = The human body contains many elements from the periodic table. It is mostly composed of oxygen and carbon, but trace elements also have a significant role. The table gives the mass, in kilograms, of some elements found in the human body, based on a 70.0 kg person. Atomic number Element Mass (kg) 8 oxygen 45.2 6. carbon 12.6 1 hydrogen 7.0 7 nitrogen 2.2 20 calcium 1.3 15 phosphorus 0.78 19 potassium 0.25 16 sulfur 0.18 others 0.60 total 70.0 Nitrogen makes up what fraction of the mass of the human body? 20 calcium 1.3 15 phosphorus 0.78 19 potassium 0.25 16 sulfur 0.18 others 0.60 total 70.0 Nitrogen makes up what fraction of the mass of the human body? fraction of N = Nitrogen makes up what percentage of the mass of the human body? %N = What is the mass of phosphorus in a 47 kg person? mass of P =
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A sample containing carbon (atomic mass 12 u), oxygen (16 u), and an unknown element is placed in a mass spectrometer. The ions all have the same charge and are accelerated through the same potential...
-
The element carbon has atomic number 6, and so it has a total of six electrons. Two of these electrons are in the 1s level. The four electrons in the 2s and 2p levels (the valence shell) are the...
-
Calcium is the most abundant mineral in the human body and has several important functions. Most body calcium is stored in the bones and teeth, where it functions to support their structure....
-
A mutual fund has 20 stocks in its portfolio. On a given day 9 stocks move up, 3 stay the same, and 8 move down. In how many ways could this happen?
-
1. Why did the court reject the plaintiffs breach of contract claim? 2. What is unjust enrichment, and why was that claim denied by the court? 3. Defects discovered in the Michelin tires to be used...
-
One solenoid is centered inside another. The outer one has a length of 50.0 cm and contains 6750 coils, while the coaxial inner solenoid is 3.0 cm long and 0.120 cm in diameter and contains 15 coils....
-
When placed between the poles of a horseshoe magnet as shown in Figure 28.12, does a rectangular current loop experience a torque? If so, in which direction does the loop rotate? Data from Figure...
-
Swisscom AG, the principal provider of telecommunications in Switzerland, prepares consolidated financial statements in accordance with International Financial Reporting Standards (IFRS). Until 2007,...
-
6. Given below are the oxides : Na2O, As2O3, N2O, NO and ChO7 Number of amphoteric oxides is: (a) 0 (b) 1 7. Match List -I with List - II: List-I List-II (A) Sphalerite (I) FeCO3 (B) Calamine (II)...
-
Christo Manufacturing Company incurs the following manufacturing cost items during the month of March 2022: (1.1) Assembly line wages (1.2) Raw materials used directly in products (1.3) Depreciation...
-
K Kaye deposited $10,000 into a savings account today. For how long can $680 be withdrawn from the account at the end of every year starting one year from now if interest is 6.5% compounded annually?...
-
Blue, Inc. earns $25M of taxable income through its U.S. operations and $10M through its wholly-owned French subsidiary. The French subsidiary did not pay any dividends to Blue during the year. What...
-
Liz is preparing a strategic plan outlining the company's decision to discontinue an existing product. What is one component that might be found in this type of plan?
-
A publisher received a 24-months subscriptions from a subscriber for $216. What are the entries to recognize the revenue after the first monthly subscription is delivered to the subscriber?
-
A client of yours has decided to take their CPP retirement pension early at the age of 60. Assuming they are entitled to a CPP Pension of $1,306.58 at normal retirement age, what will their pension...
-
The chefs want to make mayonnaise, using raw unpasteurised eggs. What is the maximum time they can keep the mayonnaise, even if it's kept refrigerated and why? Stuck? Please revisit Key factors in...
-
When Starbucks management decided to phase out plastic straws in 2018, they were considering the ______ impact of their actions as a company. A) community B) branding C) financial D) environmental
-
Imagine that your best friend knows you are taking a psychology course and wonders what psychology is all about. How would you define psychology for your friend? Write an essay on the discipline of...
-
If the bus in Problem 4 were to slow to a "mere" 10% of the speed of light, show that you would measure the passenger's catnap to last slightly more than 5 minutes.
-
The human body can maintain its customary temperature of 37C on a day when the temperature is above 40C. How is this done?
-
Does a sonic boom occur at the moment when an aircraft exceeds the speed of sound? Explain.
-
Please reflect on and explain the role and usefulness of the concept of SD in relation to the protection of the environment.
-
Has the concept of SD achieved the balance between all three pillars: environmental protection; economic development; and social issues?
-
How many dimensions of the PSNR principle can you enumerate? To which one does UN Resolution 1803(1962) refer?

Study smarter with the SolutionInn App


